
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.8, Problem 20P
Explain why, when a small amount of glycerol is added to the reaction mixture of toluene- 2,6–diisocyanate and ethylene glycol during the synthesis of polyurethane foam, a much stiffer foam is obtained.
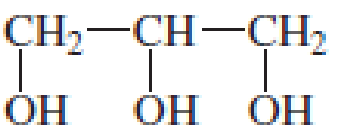
glycerol
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Account for the greater amount of internal pressure in the aspirin extraction versus the ß- naphthol extraction.Why does the sequence for extracting the diethyl ether solution of aspirin acid, naphthalene, and ß-naphthol start with aqueous bicarbonate and follow with aqueous hydroxide rather than the reverse order?
The preparation of Wittig reagents requires a strong base like butyllithium. Sodium methoxide, used in this reaction to prepare the Horner-Emmons-Wittig reagent, is a weaker base than butyllithium. Suggest a reason why this weaker base is sufficient to prepare the Horner-Emmons-Wittig reagent.
For extraction with ethyl-4-aminobenzoate and naphthalene using dichloromethane as the solvent, how does one separate the two compounds from one another?
Chapter 15 Solutions
Essential Organic Chemistry, Global Edition
Ch. 15.2 - Prob. 1PCh. 15.2 - Prob. 2PCh. 15.2 - Prob. 3PCh. 15.2 - Prob. 4PCh. 15.2 - List the following groups of monomers in order...Ch. 15.2 - List the following groups of monomers in order...Ch. 15.2 - Prob. 7PCh. 15.2 - Why does methyl methacrylate not undergo cationic...Ch. 15.2 - Which monomer and which type of initiator would...Ch. 15.2 - Prob. 10P
Ch. 15.2 - Prob. 11PCh. 15.5 - Draw a short segment of gutta-percha.Ch. 15.5 - Prob. 13PCh. 15.6 - Prob. 14PCh. 15.8 - Prob. 15PCh. 15.8 - Prob. 16PCh. 15.8 - Prob. 17PCh. 15.8 - a. Propose a mechanism for the formation of the...Ch. 15.8 - Propose a mechanism for the formation of Melmac.Ch. 15.8 - Explain why, when a small amount of glycerol is...Ch. 15.10 - Prob. 21PCh. 15 - Draw short segments of the polymers obtained from...Ch. 15 - Prob. 23PCh. 15 - Draw the structure of the monomer or monomers used...Ch. 15 - Draw short segments of the polymers obtained from...Ch. 15 - Prob. 26PCh. 15 - Prob. 27PCh. 15 - Prob. 28PCh. 15 - A particularly strong and rigid polyester used for...Ch. 15 - Prob. 30PCh. 15 - Prob. 31PCh. 15 - Prob. 32PCh. 15 - Prob. 33PCh. 15 - Prob. 34PCh. 15 - Prob. 35PCh. 15 - Delrin (polyoxymethylene) is a tough...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain why the addition of a small amount of glycerol to the polymerization mixture gives a stiffer urethane foam.arrow_forwardin a liquid-liquid extraction, why is ethyl acetate a good initial solvent to dissolve a mixture of benzoic acid, 4-bromoacetophenone and ethyl 4-aminobenzoate? the extraction solvent is sodium bicarbonate.arrow_forwardWhy is the 4-methoxyphenyl acetate organic layer subjected to washes with saturated sodium bicarbonate solution, water and brine when carrying out a separatory funnel extraction?arrow_forward
- Regarding an organic chemistry laboratory experiment: a. Why is it necessary to isolate eugenol from cloves using (direct) steam distillation rather simple distillation? b. In this experiment, why does eugenol distill at a temperature far below its boiling point? c. Why is diethyl ether used to extract eugenol from the distillate rather than ethanol?arrow_forwardwhat will happen to the reaction of acetone in the sodium nitroprusside Test and in the iodoform test? what will happen if liquid Benzaldehyde is exposed into the air?arrow_forwardHow would you expect the melting point of the polyvinyl alcohol would compare to that of the cross-linked polyvinyl alcohol. Explain how you arrived at your explanation?arrow_forward
- Why is Acetic Anhydride soluble in sulfuric acid?arrow_forwardIn addition to the normal dangers in using concentrated hydrochloric acid, what additional dangers are presented in mixing it with ice and what precautions should be taken? What gas is evolved when hydrochloric acid is added to the borohydride reduction with benzophenone?arrow_forwardIn the tests with sodium iodide in acetone and silver nitrate in ethanol, why should 2-bromobutane react faster than 2-chlorobutane?arrow_forward
- Why is it necessary to store the prepared lysates in a very low temperature?arrow_forwardWhy is the increase in regioselectivity for isopropyl benzene less than the increase in regioselectivity for toluene when you add a co solvent to both of them?arrow_forwardIn a paragraph form provide the experimental procedure of 3-benzoylpropionic acid reaction of Clemmenson, which results in 4-phenyl butanoic acid. Clemmensen reaction is observed in the presence of zinc amalgam and hydrochloric acidarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY