
Concept explainers
Propose structures for
(a) C9H12; gives only one C9H11Br product on substitution of a hydrogen on the aromatic ring with bromine
(b) C10H14; gives only one C10H13Cl product on substitution of a hydrogen on the aromatic ring with chlorine
(c) C8H10; gives three C8H9Br products on substitution of a hydrogen on the aromatic ring with bromine
(d) C10H14; gives two C10H13Cl products on substitution of a hydrogen on the aromatic ring with chlorine
a) C9H12; gives only one C9H11Br product on substitution of a hydrogen on the aromatic ring with bromine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen on the aromatic ring with bromine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring.
To propose:
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen on the aromatic ring with bromine.
Answer to Problem 23AP
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen atom on the aromatic ring with bromine is
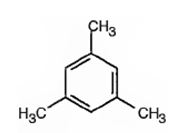
Explanation of Solution
The molecular formula of the compound is C9H12. Benzene ring accounts for six carbons. When the three carbons remaining are arranged as three methyl groups alternatively on the benzene ring the three hydrogens on the other three carbons become equivalent. Hence upon bromination, the hydrocarbon gives only one monosubstituted product.
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen atom on the aromatic ring with bromine is

b) C10H14; gives only one C10H13Cl product on substitution of a hydrogen on the aromatic ring with chlorine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product C10H13Cl on substitution of a hydrogen on the aromatic ring with chlorine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring. Only one monosubstituted product will be obtained only if all the hydrogen atoms in the aromatic ring are equivalent.
To propose:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product C10H13Cl on substitution of a hydrogen on the aromatic ring with chlorine is to be given.
Answer to Problem 23AP
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product, C10H13Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is
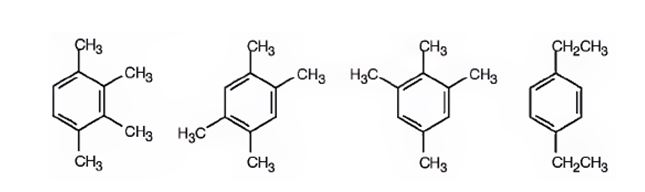
Explanation of Solution
The molecular formula of the hydrocarbon is C10H14. Benzene ring accounts for six carbons. The remaining four carbons can be arranged as four methyl groups or as two ethyl groups on the benzene ring in four different ways as shown such that remaining hydrogen atoms become equivalent. Hence upon chlorination, the hydrocarbon will give only one monosubstituted product.
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product, C10H14Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is

c) C8H10; gives three C8H9Br products on substitution of a hydrogen on the aromatic ring with bromine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three monobromination products. C8H9Br, on substitution of a hydrogen on the aromatic ring with bromine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring.
To propose:
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three products, C8H9Br, on substitution of a hydrogen on the aromatic ring with bromine.
Answer to Problem 23AP
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three monobromination products C9H11Br on substitution of a hydrogen atom on the aromatic ring with bromine is
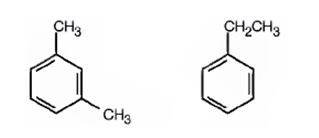
Explanation of Solution
The molecular formula of the hydrocarbon is C8H10. Benzene ring accounts for six carbons. When the two carbons remaining are arranged as two methyl groups meta to each other or as an ethyl group on the benzene ring hydrogens on the other carbon atoms in the ring classify themselves into three different groups. Hence upon bromination, the hydrocarbon gives three monosubstituted products.
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three monobromination products, C9H11Br, on substitution of a hydrogen atom on the aromatic ring with bromine is

d) C10H14; gives two C10H13Cl products on substitution of a hydrogen atom on the aromatic ring with chlorine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives two monochlorination products C10H13Cl on substitution of a hydrogen atom on the aromatic ring with chlorine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring.
To propose:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives two products, C10H13Cl, on substitution of a hydrogen on the aromatic ring with chlorine.
Answer to Problem 23AP
Possible structure for the hydrocarbon with molecular formula C10H10 that gives two monobromination products, C10H13Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is
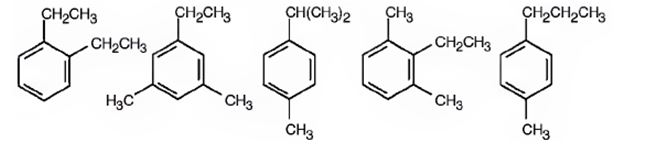
Explanation of Solution
The molecular formula of the compound is C10H14. Benzene ring accounts for six carbons. The four carbons remaining can be arranged as two ethyl groups or as an ethyl & two methyl groups or as propyl & methyl groups or as isopropyl & methyl groups on the benzene ring as shown so that the hydrogens on the other carbon atoms in the ring classify themselves into two different groups. Hence upon chlorination, the hydrocarbon gives two monosubstituted products.
A possible structure for the hydrocarbon with molecular formula C10H10 that gives two monobromination products, C10H13Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is

Want to see more full solutions like this?
Chapter 15 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Treatment of a hydrocarbon A (molecular formula C9H18) with Br2 in the presence of light forms alkyl halides B and C, both having molecular formula C9H17Br. Reaction of either B or C with KOC(CH3)3 forms compound D (C9H16) as the major product. Ozonolysis of D forms cyclohexanone and acetone. Identify the structures of A–D.arrow_forwardFor following substituted benzenes: [1] C6H5Br; [2] C6H5CN; [3] C6H5OCOCH3: Does the substituent activate or deactivate the benzene ring inelectrophilic aromatic substitution?arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forward
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward(b) Propose a synthesis of (2,2-dimethylpropyl)benzene from benzene.arrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents.(a) HNO3, H2SO4 (b) Br2, FeBr3 (c) CH3CH2COCl, AlCl3(d) isobutylene and HFarrow_forward
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents.(a) HNO3, H2SO4arrow_forwardThe following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardSynthesize the following compound from benzonitrile (C6H5CN):arrow_forward
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with HNO3, H2SO4 (b) Br2, FeBr3arrow_forward4) Aromatic compounds are among the most abundant and versatile in nature. From a synthetic point of view, these compounds, despite their stabilities, are quite useful and can undergo reactions under special conditions and by specific mechanisms, such as the Electrophilic Aromatic Substitution (SAE) and the Nucleophilic Aromatic Substitution (SNAr). Based on this, please answer the following items: (b) How would you prepare the following compounds starting from benzene? Explain the second in a different wayarrow_forward1. At what position and on what ring would you expect the following substances to undergo electrophilic substitution? (b) CH3 Br lel CH3 2. Rank the compounds in each group according to their reactivity toward electrophilic substitution. (a) Chlorobenzene, o-dichlorobenzene, benzene (b) p-Bromonitrobenzene, nitrobenzene, phenol (c) Fluorobenzene, benzaldehyde, 0-xylene (d) Benzonitrile, p-methylbenzonitrile, p-methoxybenzonitrilearrow_forward
