
(a)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
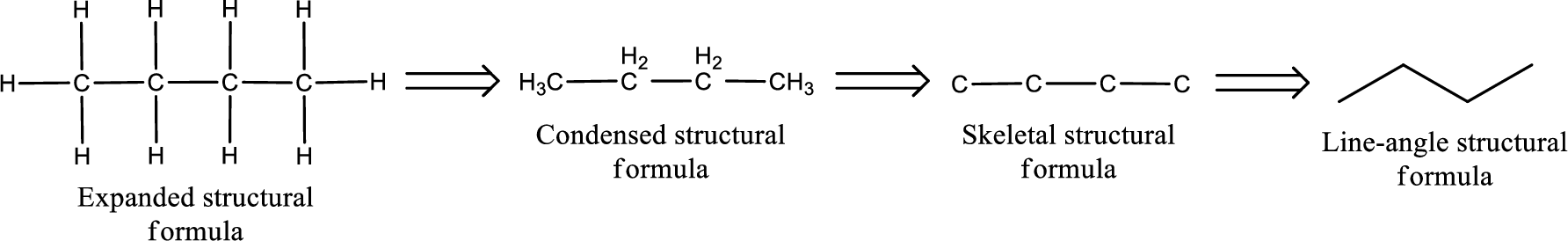
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
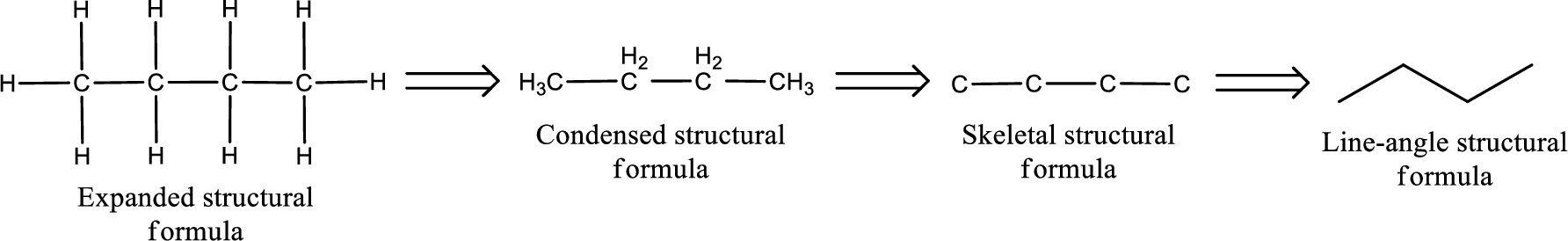
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(a)
Answer to Problem 16.29EP
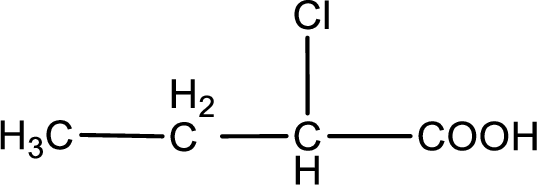
The condensed structural formula is,
Explanation of Solution
Given description about the carboxylic acid is
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a four carbon monocarboxylic acid. The chlorine atom is substituted in the alpha carbon atom. Alpha carbon atom in carboxylic acid is the one that is attached to the carboxyl group. Therefore, the structure can be drawn as shown below,
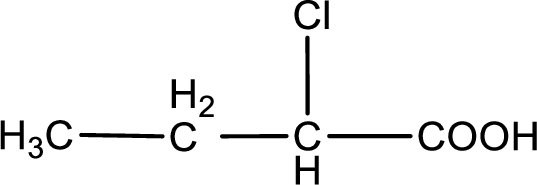
The condensed structural formula for the given carboxylic acid is drawn.
(b)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
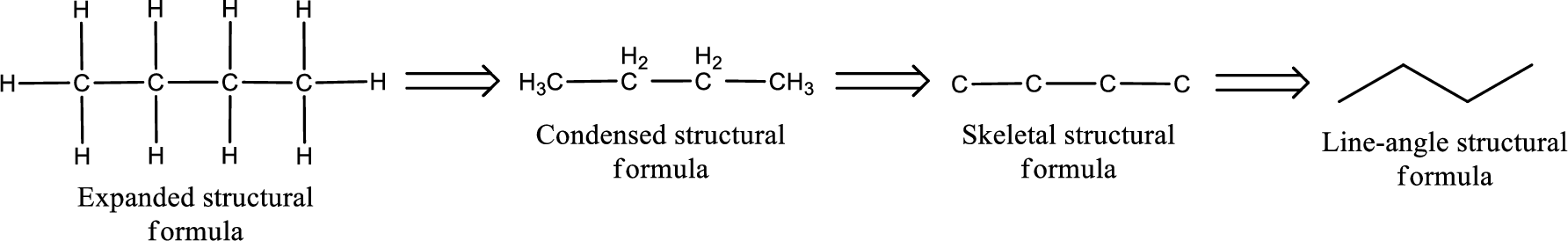
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(b)
Answer to Problem 16.29EP
The condensed structural formula is,
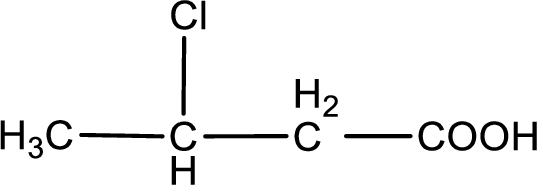
Explanation of Solution
Given description about the carboxylic acid is
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a four carbon monocarboxylic acid. The chlorine atom is substituted in the beta carbon atom. Beta carbon atom in carboxylic acid is the one that is the second carbon atom from the carboxyl group. Therefore, the structure can be drawn as shown below,
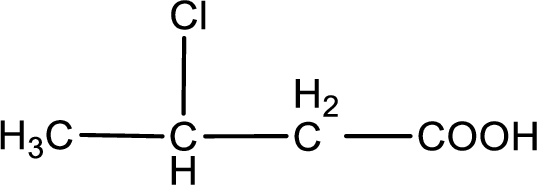
The condensed structural formula for the given carboxylic acid is drawn.
(c)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(c)
Answer to Problem 16.29EP
The condensed structural formula is,
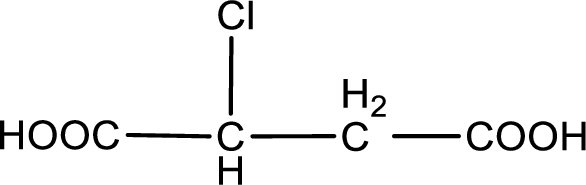
Explanation of Solution
Given description about the carboxylic acid is chlorosuccinic acid.
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a four carbon dicarboxylic acid. The chlorine atom is substituted in the either of the two carbon atoms as both the carbon atoms present between the two carboxyl groups are identical. Therefore, the structure can be drawn as shown below,
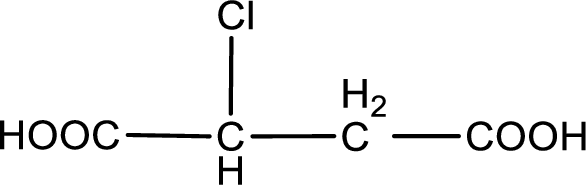
The condensed structural formula for the given carboxylic acid is drawn.
(d)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(d)
Answer to Problem 16.29EP
The condensed structural formula is,
Explanation of Solution
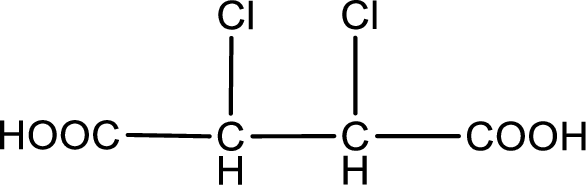
Given description about the carboxylic acid is
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a four carbon dicarboxylic acid. The chlorine atom is substituted in the alpha carbon atom and beta carbon atom. Alpha carbon atom in carboxylic acid is the one that is attached to the carboxyl group and the next carbon attached to the alpha carbon atom is the beta carbon atom. Therefore, the structure can be drawn as shown below,
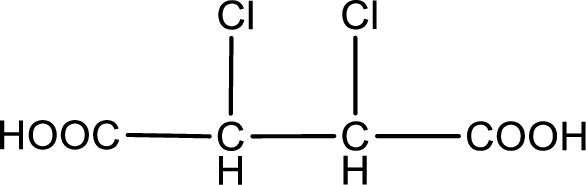
The condensed structural formula for the given carboxylic acid is drawn.
Want to see more full solutions like this?
Chapter 16 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Draw the structure of the tripeptide alanylglycylvaline and determine its name using three-letter abbreviations.arrow_forwardIs D-2-deoxygalactose the same chemical as D-2-deoxyglucose? Draw and explainarrow_forwardIn the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forward
- Provide compounds that fit the following descriptions:(a) Two amines that are gases at room temperature(b) A heterocyclic amine(c) A compound with an amine group on an aromatic ringarrow_forwardThe components of poison ivy and poison oak that produce the characteristic itchy rash are catechols substituted with long-chain alkylgroups. If you were exposed to poison ivy, which of the treatments below would you apply to the affected area? Justify your choice.(a) Wash the area with cold water.(b) Wash the area with dilute vinegar or lemon juice.(c) Wash the area with soap and water.(d) Wash the area with soap, water, and baking soda (sodium bicarbonate).arrow_forwardDefine the following terms: a. reducing sugar b. alditol c. enediol d. acetal e. ketalarrow_forward
- A) Describe the glycosidic bond (using standard convention) indicated by “Arrow a.” B) Draw the open chain Fischer projection formula of the monosaccharide labeled “B” C) Describe the glycosidic bond (as in question A) indicated by “Arrow b.”arrow_forwardWrite one Chemical difference and similarity between an amine and an amidearrow_forwardwhat is the conjugate acid for the following reaction? HC2HO4 + H2O <--> H3O+ + C2HO4- A)C2HO4- B) HC2HO4 C)H2O D) H3O+arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





