
ORGANIC CHEMISTRY LSLF W/CONNECT >BI<
5th Edition
ISBN: 9781260293609
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.29P
Name each diene and state whether the ball-and-stick model shows the diene in the
a. 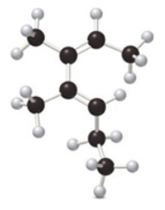 b.
b. 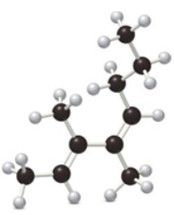
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which group in each pair is assigned the higher priority?
a. – CH3, – CH2CH3
b. – I, – Br
c. – H, – D
d. – CH2Br, – CH2CH2Br
e. – CH2CH2Cl, – CH2CH(CH3)2
f. – CH2OH, – CHO
Which diene is more reactive in a Diels–Alder reaction? CH2=CHCH=CHOCH3 or CH2=CHCH=CHCH2OCH3
Label each diene as reactive or unreactive in a Diels–Alder reaction
Chapter 16 Solutions
ORGANIC CHEMISTRY LSLF W/CONNECT >BI<
Ch. 16 - Prob. 16.1PCh. 16 - Prob. 16.2PCh. 16 - Problem 16.3 Draw a second resonance structure for...Ch. 16 - Prob. 16.4PCh. 16 - Problem 16.5 Farnesyl diphosphate is synthesized...Ch. 16 - Prob. 16.6PCh. 16 - Prob. 16.7PCh. 16 - Prob. 16.8PCh. 16 - Problem 16.9 Determine the hybridization of the...Ch. 16 - Problem 16.10 Draw the structure consistent with...
Ch. 16 - Problem 16.11 Neuroprotectin D1 (NPD1) is...Ch. 16 - Problem 16.12 Using hybridization, predict how the...Ch. 16 - Problem 16.13 Use resonance theory to explain why...Ch. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - Problem 16.16 Draw the products formed when each...Ch. 16 - Problem 16.17 Draw a stepwise mechanism for the...Ch. 16 - Prob. 16.18PCh. 16 - Problem 16.19 Draw the product formed when each...Ch. 16 - Prob. 16.20PCh. 16 - Prob. 16.21PCh. 16 - Problem 16.22 Rank the following dienophiles in...Ch. 16 - Prob. 16.23PCh. 16 - Prob. 16.24PCh. 16 - Problem 16.25 What diene and dienophile are needed...Ch. 16 - Prob. 16.26PCh. 16 - Problem 16.27 Which compound in each pair absorbs...Ch. 16 - Prob. 16.28PCh. 16 - 16.29 Name each diene and state whether the...Ch. 16 - Prob. 16.30PCh. 16 - 16.31 Which of the following systems are...Ch. 16 - 16.32 Draw all reasonable resonance structures for...Ch. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - 16.35 Explain why the cyclopentadienide anion A...Ch. 16 - Prob. 16.36PCh. 16 - 16.37 Draw the structure of each compound.
a. in...Ch. 16 - Prob. 16.38PCh. 16 - 16.39 Label each pair of compounds as...Ch. 16 - Prob. 16.40PCh. 16 - 16.41 Draw the products formed when each compound...Ch. 16 - Prob. 16.42PCh. 16 - 16.43 Treatment of alkenes A and B with gives the...Ch. 16 - 16.44 Draw a stepwise mechanism for the following...Ch. 16 - Prob. 16.45PCh. 16 - 16.46 Explain, with reference to the mechanism,...Ch. 16 - Prob. 16.47PCh. 16 - Prob. 16.48PCh. 16 - Prob. 16.49PCh. 16 - Prob. 16.50PCh. 16 - Prob. 16.51PCh. 16 - Prob. 16.52PCh. 16 - 16.53 Diels–Alder reaction of a monosubstituted...Ch. 16 - Prob. 16.54PCh. 16 - 16.55 Devise a stepwise synthesis of each compound...Ch. 16 - Prob. 16.56PCh. 16 - 16.57 A transannular Diels–Alder reaction is an...Ch. 16 - Prob. 16.58PCh. 16 - Draw a stepwise mechanism for the following...Ch. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - 16.65 The treatment of isoprene with one...Ch. 16 - 16.66 The treatment of with forms B (molecular...Ch. 16 - Prob. 16.67PCh. 16 - Prob. 16.68PCh. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Prob. 16.73PCh. 16 - Prob. 16.74PCh. 16 - Prob. 16.75P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In 4+2 cycloaddition, ENDO stereochemistry is preferred because: endo stereochemistry allows the diene to adopt s-cis conformation. endo stereochemistry results in less steric hindrance. the transition state leading to endo stereochemistry is higher energy. it allows for additional π to π overlap between the diene and the dienophile.arrow_forward(a) What product(s) are formed when the E isomer of C6H5CH = CHC6H5 is treated with Br2, followed by one equivalent of KOH? Label the resulting alkene(s) as E or Z. (b) What product(s) are formed when the Z isomer of C6H5CH = CHC6H5 is subjected to the same reaction sequence? (c) How are the compounds in parts (a) and (b) related to each other?arrow_forwardThe central carbon atom of an allene is a member of two double bonds, and ithas an interesting orbital arrangement that holds the two ends of the molecule at rightangles to each other.(a) Draw an orbital diagram of allene, showing why the two ends are perpendicular.(b) Draw the two enantiomers of penta-2,3-diene. A model may be helpfularrow_forward
- Draw the products formed when the diene is treated with O3 followed by CH3SCH3.arrow_forwardWith reference to diene A:a. What product is formed when A undergoes a [2 + 2] cycloaddition?b. What product is formed when A undergoes a [3,3] sigmatropic rearrangement?arrow_forwardDraw an energy diagram for an E2 reaction: (CH3)3CBr + −OH →(CH3)2C=CH2 + H2O + Br−arrow_forward
- Which dienophile in each pair is more reactive in a Diels–Alder reaction? 1. CH2=CH=OCH or CH2=CHCH2=OCH 2. CH2=CH=OCH or CH2=CHCH3arrow_forwarda. How many s bond orbitals are available for overlap with the vacant p orbital in 1. the isobutyl cation? 2. the n-butyl cation? 3. the sec-butyl cation? b. Which of the carbocations in part a is most stable?arrow_forwardAddition of HBr to allene (CH2=C=CH2) forms 2-bromoprop-1-ene ratherthan 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed froman allylic carbocation. Considering the arrangement of orbitals in theallene reactant, explain this result.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License