
Concept explainers
RECALL Define the following terms: polysaccharide, furanose, pyranose, aldose, ketose, glycosidic bond, oligosaccharide, glycoprotein.
Interpretation:
The given terms are to be defined.
Concept introduction:
A long chain of monosaccharides linked by glycosidic bonds is known as a polysaccharide. A five-membered cyclic ring of sugar is furanose and a six-membered cyclic ring is pyranose. A monosaccharide containing an aldehydic group is called an aldose, and if it contains a ketonic group then it is called a ketose. The hydroxyl
Answer to Problem 1RE
Solution:
Polysaccharide: It is a large molecule made up of a large number of monosaccharides that are bound together with glycosidic linkages.
Pyranose: It is a six-membered cyclic sugar consisting of five carbon atoms along with one oxygen atom.
Furanose: It is a five-membered cyclic sugar consisting of four carbon atoms along with one oxygen atom.
Aldose: It is a monosaccharide (a simple sugar) that contains an aldehydic group
Ketose: It is a monosaccharide (a simple sugar) that contains a ketonic group
Glycosidic bond or glycosidic linkage: It is a type of covalent bond that joins one carbohydrate molecule to another carbohydrate molecule.
Oligosaccharide: It is a saccharide polymer that contains a small number of monosaccharides, typically 3–10 monosaccharides molecules.
Glycoproteins: These proteins consist of oligosaccharides that are covalently attached to proteins (or amino acids).
Explanation of Solution
Given information: Polysaccharides, pyranose, furanose, aldose, ketose, glycoside bond, oligosaccharide, and glycoprotein.
Polysaccharide: It is a polymer that consists of a large number of monosaccharides, typically around 20 or more. They are of two types, namely, homopolysaccharides, which consist of a single type of single monosaccharide unit, and heterpolysaccharides, which consist of different monosaccharide units. For example, glycogen, a homopolysacccharide, forms glucose units.
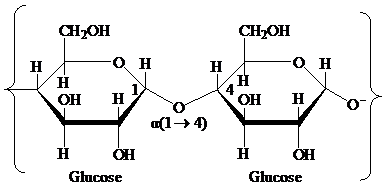
Pyranose: The chemical structure of pyranose contains a six-membered ring, which includes five carbon atoms and one oxygen atom. The formation of pyranose takes place by the reaction of alcoholic group of carbon-5 of an open chain with the aldehydic group of carbon-1 of a similar chain.
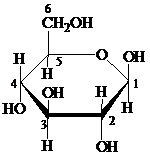
Furanose: The chemical structure of furanose contains a five-membered ring that includes four carbon atoms and one oxygen atom. The formation of pyranose takes place by the reaction of alcoholic group of carbon-4 of an open chain with the aldehydic group of carbon-1 of a similar chain.
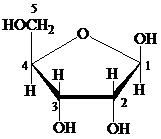
Aldose: This is a class of carbohydrates that has an aldehydic group and is known as polyhydroxy aldehydes. Aldoses are also called reducing sugars for they act as a reducing agent because of the presence of the aldehydic group. They reduce Tollen’s reagent. For example, glucose.
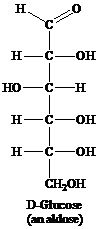
Ketose: This is a class of carbohydrates that have a ketonic group and is also known as polyhydroxy ketones. They are not reducing sugars but they tautomerize themselves to aldoses before acting as a reducing sugar. For example, fructose
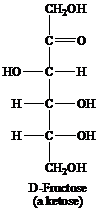
Glycosidic bond: It is a covalent bond that joins a carbohydrate to another carbohydrate or functional group of any other molecule. A substance containing a glycosidic bond is called a glycoside. For example, the glycosidic linkage of two glucose molecules is shown in the following figure:
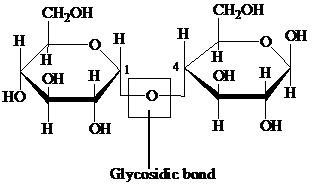
Oligosaccharides: Such carbohydrates contain 3–10 simple sugars that are linked together. They are naturally found in plants and onions. They are soluble fibers and hence easy to digest. Most common examples are sucrose, maltose, and lactose.

Glycoprotein: It is a type of protein that has a carbohydrate attached to it. This carbohydrate is an oligosaccharide that is covalently attach to a polypeptide chain of protein. It is usually found in aloe vera, brown rice, meat, and so on. They play an important function in the reproduction, immune system, hormone balance, and protection of cells.
All the terms have been defined, along with their structures being described.
Want to see more full solutions like this?
Chapter 16 Solutions
Biochemistry - With Access
- RECALL Is it possible to form cyclic peptides without bonds between side chains of the component amino acids?arrow_forwardREFLECT AND APPLY Compare the information in the sequence of monomers in a polysaccharide with that in the sequence of amino acid residues in a protein.arrow_forwardRECALL What are glycoproteins? What are some of their biochemical roles?arrow_forward
- RECALL What types of macromolecules are usually separated on agarose electrophoresis gels?arrow_forwardRECALL What is a suicide substrate? Why are they important?arrow_forwardRECALL How can gel-filtration chromatography be used to arrive at an estimate of the molecular weight of a protein?arrow_forward
- REFLECT AND APPLY Nucleic acids are polymers of just four different monomers in a linear arrangement. How many different sequences are available if one makes a polymer with only 40 monomers? How does this number compare with Avogadros number?arrow_forwardRECALL What is a chaperone?arrow_forwardRECALL Are amino acids other than the usual 20 amino acids found in proteins? If so, how are such amino acids incorporated into proteins? Give an example of such an amino acid and a protein in which it occurs.arrow_forward
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
