
(a)
Interpretation:
The structural formula for the product formed by the treatment of Butanal with
Concept Introduction:
Butanal is a compound with four carbon atoms and −CHO group.
The structure of Butanal is as follows:
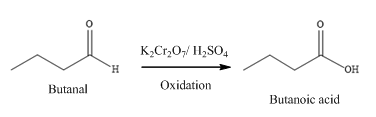
(b)
Interpretation:
The structural formula for the product formed by the treatment of Butanal with
Concept Introduction:
Butanal is a compound with four carbon atoms and −CHO group.
The structure of Butanal is as follows.
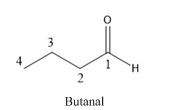
(c)
Interpretation:
The structural formula for the product formed by the treatment of Butanal with Tollen’s reagent should be drawn.
Concept Introduction:
Butanal is an aldehyde and the compound which have four carbons.
The structure of Butanal is as follows.
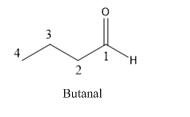
(d)
Interpretation:
The structural formula for the product formed by the treatment of Butanal with
Concept Introduction:
Butanal is an aldehyde and the compound which have four carbons.
The structure of Butanal is as follows.
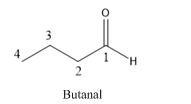
Trending nowThis is a popular solution!

Chapter 16 Solutions
Introduction To General, Organic, And Biochemistry
- 17-33 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Pentanal and 2-pentanone (b) 2-Pentanone and 2-pentanolarrow_forward17-35 Suppose that you take a bottle of benzaldehyde (a liquid, bp 179°C) from a shelf and find a white solid in the bottom of the bottle. The solid turns litmus red; that is, it is acidic. Yet aldehydes are neutral compounds. How can you explain these observations?arrow_forward17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward
- 17-36 Explain why the reduction of an aldehyde always gives a primary alcohol and the reduction of a ketone always gives a secondary alcohol.arrow_forward2 (Chemical Connections 19A) Locate the ester group in pyrethrin I and draw a structural formula for chrysanthemic acid, the carboxylic acid from which this ester is derived.arrow_forward18-48 4-Aminobenzoic acid is prepared from benzoic acid by the following two steps. Show reagents and experimental conditions to bring about each step.arrow_forward
- 7 What type of structural feature do the anhydrides of phosphoric acid have in common with carboxylic acids?arrow_forward17-34 Explain why liquid aldehydes are often stored under an atmosphere of nitrogen rather than in air.arrow_forward17-28 Show how acetaldehyde can form hydrogen bonds with water.arrow_forward
- 17-69 Propanal (bp 49°C) and 1-propanol (bp 97°C) have about the same molecular weight, yet their boiling points differ by almost 50°C. Explain this fact.arrow_forward17-72 The following molecule is an enediol; each carbon of the double bond carries an —OH group. Draw structural formulas for the hydroxyketone and the a-hydroxyaldehyde with which this enediol is in equilibrium.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

