
Concept explainers
Compounds W and X are isomers; they have the molecular formula
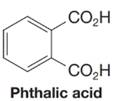
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Chemistry: Structure and Properties
Living by Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Organic Chemistry As a Second Language: Second Semester Topics
Organic Chemistry (8th Edition)
- A compound (C7H14O) has a strong peak in its IR spectrum at 1710 cm–1. Its 1H NMR spectrum consists of three singlets in the ratio 9:3:2 at δ 1.0, 2.1, and 2.3, respectively. Identify the compound.arrow_forwardA hydrocarbon, compound B, has molecular formula C6H6, and gave an NMR spectrum with two signals: delta 6.55 pm and delta 3.84 pm with peak ratio of 2:1. When warmed in pyridine for three hr, compound B quantitatively converts to benzene. Mild hydrogenation of B yielded another compound C with mass spectrum of m/z 82. Infrared spectrum showed no double bonds; NMR spectrum showed one broad peak at delta 2.34 ppm. With this information, address the following questions. a) How many rings are in compound C? b) How many rings are probably in B? How many double bonds are in B? c) Can you suggest a structure for compounds B and C? d) In the NMR spectrum of B, the up-field signal was a quintet, and the down field signal was a triplet. How must you account for these splitting patterns?arrow_forwardThere are several isomeric alcohols and ethers of molecular formula C5H12O. Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.92 (t, 7.8 Hz, 3 H), 1.20 (s, 6H), 1.49 (q, 7.8 Hz, 2H), 1.85 (s, 1H) ppm Isomer B: δ = 1.19 (s, 9 H), 3.21 (s, 3H) ppmarrow_forward
- Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)arrow_forwardCompound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forwardA compound has the molecular formula C6H12O2. Its IR spectrum shows a strong absorption band near 1740 cm-1. Its 1H NMR spectrum consists of two singlets at δ 1.2 and δ 3.6. Which is the most likely structure of the compound?arrow_forward
- There are several isomeric alkanes of molecular formula C6H14.Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (s, 9H), 1.22 (q, 2H) ppmarrow_forwardCompound X (molecular formula C10H12O) was treated with NH2NH2, −OH to yield compound Y (molecular formula C10H14). Based on the 1HNMR spectra of X and Y given below, what are the structures of X and Y?arrow_forwardAn unknown compound C3H2NCl shows moderately strong IR absorptions around 1650 cm-1 and 2200 cm-1. Its NMR spectrum consists of two doublets (J = 14 Hz) at δ 5.9 and δ 7.1. Propose a structure consistent with this data?arrow_forward
- Compounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm−1 . The 1H NMR spectra of Y and Z are given below. Propose structures for Y and Z.arrow_forwardWhen a compound with molecular formula C11H14O2 undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following 1H NMR spectrum. Identify the compound.arrow_forwardWhen compound X (C15H17N) is treated with bezenesulfonyl chloride and aqueous potassium hydroxide, no apparent change occurs. Acidification of the mixture gives a clear solution. Propose a structure for X based on the given 1H NMR spectra. Explain your answer by interpreting the spectraarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
