
Concept explainers
Interpretation:
The new nucleus after the emission of a positron needs to be determined.
Concept introduction:
A proton is turned into a neutron and a positron (with a positive charge) leaves the nucleus.
Positron emission happens when a nucleus emits a positron
Answer to Problem 51UTC
Solution:
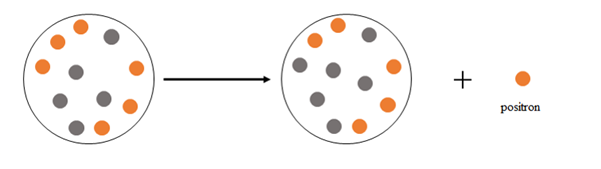
Explanation of Solution
Positron emission happens when a nucleus emits a positron
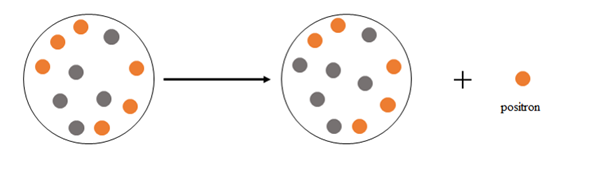
At the beginning there are 6 protons and 5 neutrons, after the emission there are 5 protons and 6 neutrons.
Thus, positron emission happens when a nucleus emits a positron
Want to see more full solutions like this?
Chapter 16 Solutions
Basic Chemistry, Loose-leaf Edition (6th Edition)
- co-60 is used for radiation therapy as implants and as external source of radiation exposure. The half life of Co-60 is 5,272 years. how much of a 2.000 mg sample will remain after 21,088 years? Please show work.arrow_forwardStrontium-90 is a dangerous by-product of atomic testing because it mimics the action of calcium in the body. It decay in two B-emissions to give zirconium-90. Write a balanced nuclear equation for the overall decay of Sr-90. Calculate ◇m in grams when one mole of Sr-90 decays to Zr-90. How much energy (in KJ) is given off by the decay of 6.50 mg of Sr-90?arrow_forward- Give reason for 5- Isotopes have ability to undergo nuclear reactions which are very important in medical applications. 6- Radiation source in AAS device is hallow cathode lamp. 7- The PH of a solution Mg(OH)2 can affect a salt solubility by addition of H+ ion , pH decreasing while solubility of Mg(OH)2 will be increasing. * Short answer if you can ❤️*arrow_forward
- In the electron capture of In-110 A. The In-110 and the electron placed in the reactant side while the Cd-110 in the product side. B. The In-110 is placed in the reactant side while the Cd-110 and the electron in the product side. C. The Cd-110 and electron are placed in the reactant side while the In-110 in the product side. D. The Cd-110 is placed in the reactant side while the In-110 and the electron in the product side.arrow_forwardWhat is the energy change when the temperature of 14.0 grams of gaseous helium is decreased from 38.7 °C to 20.2 °C ?arrow_forwardIs -0.05 or 0.2 closer to 0 ?arrow_forward
- If a match contained 0.265 g of P4S3, what would be the energy released in kJ?arrow_forwardWhich has the LEAST radioactive carbon component? a. present plants b. crude oil c. whisky d. wine *Choose only one answer and explain.arrow_forwardStrontium-90 is a dangerous by-product of atomic testing because it mimics the action of calcium in the body. It decay in two β-emissions to give zirconium-90 (nuclear mass = 89.8824 g).a) Write a balanced nuclear equation for the overall decay of Sr-90.b) Calculate Δm in grams when one mole of Sr-90 decays to Zr-90.c) How much energy (in KJ) is given off by the decay of 6.50 mg of Sr-90?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

