
Principles of Physics
5th Edition
ISBN: 9781133712725
Author: SERWAY
Publisher: CENGAGE CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 53P
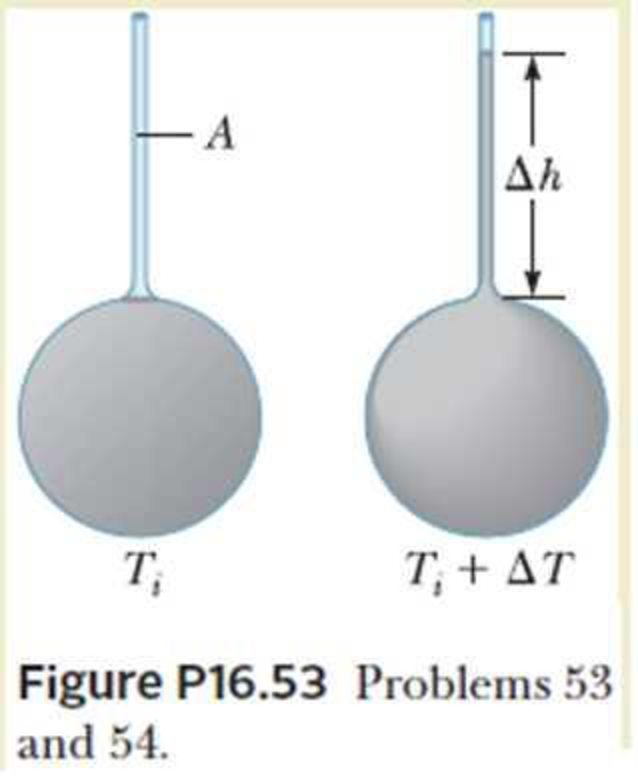
A mercury thermometer is constructed as shown in Figure P16.53. The Pyrex glass capillary tube has a diameter of 0.004 00 cm, and the bulb has a diameter of 0.250 cm. Find the change in height of the mercury column that occurs with a temperature change of 30.0°C.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A mercury thermometer is constructed
as shown in Fig. The capillary tube has a diameter of
0.004 00 cm, and the bulb has a diameter of 0.250 cm.
neglecting the expansion of the glass, find the change in
height of the mercury column with a temperature change
of 30.0°C.
T₁
A
Fig.
T, + AT,
Ah
The column of mercury in a barometer (see Figure below) has a height of 0.760 m when the pressure is one atmosphere and the
temperature is 0.0 °C. Ignoring any change in the glass containing the mercury, what will be the height of the mercury column for the
same one atmosphere of pressure when the temperature rises to 33.1 °C on a hot day? Hint: The pressure in the barometer is Pressure
pgh, and the density p changes when the temperature
changes.
Number
i
Units
……….…………ມາ
WIII |
Empty, except for a
negligible amount
of mercury vapor
(P₁ = 0 Pa)
B (P2= Atmospheric
pressure)
An air bubble has a volume of 1.60 cm3 when it is released by a submarine 115 m below the surface of a lake. What is the volume of the bubble when it reaches the surface? Assume the temperature and the number of air molecules in the bubble remain constant during its ascent.
Chapter 16 Solutions
Principles of Physics
Ch. 16.2 - Consider the following pairs of materials. Which...Ch. 16.3 - Prob. 16.2QQCh. 16.4 - A common material for cushioning objects in...Ch. 16.4 - Prob. 16.4QQCh. 16.5 - Two containers hold an ideal gas at the same...Ch. 16.6 - Prob. 16.6QQCh. 16 - Prob. 1OQCh. 16 - A cylinder with a piston holds 0.50 m3 of oxygen...Ch. 16 - A hole is drilled in a metal plate. When the metal...Ch. 16 - When a certain gas under a pressure of 5.00 106...
Ch. 16 - Prob. 5OQCh. 16 - Prob. 6OQCh. 16 - What would happen if the glass of a thermometer...Ch. 16 - Prob. 8OQCh. 16 - A gas is at 200 K. If we wish to double the rms...Ch. 16 - Prob. 10OQCh. 16 - Prob. 11OQCh. 16 - A rubber balloon is filled with 1 L of air at 1...Ch. 16 - Prob. 13OQCh. 16 - An ideal gas is contained in a vessel at 300 K....Ch. 16 - Prob. 15OQCh. 16 - Prob. 16OQCh. 16 - Prob. 17OQCh. 16 - A sample of gas with a thermometer immersed in the...Ch. 16 - Prob. 19OQCh. 16 - Prob. 1CQCh. 16 - Prob. 2CQCh. 16 - Prob. 3CQCh. 16 - A piece of copper is dropped into a beaker of...Ch. 16 - Prob. 5CQCh. 16 - Prob. 6CQCh. 16 - Prob. 7CQCh. 16 - Prob. 8CQCh. 16 - Prob. 9CQCh. 16 - Prob. 10CQCh. 16 - Prob. 11CQCh. 16 - Prob. 12CQCh. 16 - Prob. 13CQCh. 16 - Prob. 1PCh. 16 - Convert the following to equivalent temperatures...Ch. 16 - Prob. 3PCh. 16 - Prob. 4PCh. 16 - Prob. 5PCh. 16 - Prob. 6PCh. 16 - Prob. 7PCh. 16 - Prob. 8PCh. 16 - Prob. 9PCh. 16 - A sample of a solid substance has a mass m and a...Ch. 16 - Each year thousands of children are badly burned...Ch. 16 - Prob. 12PCh. 16 - Prob. 13PCh. 16 - Prob. 14PCh. 16 - The active element of a certain laser is made of a...Ch. 16 - Prob. 16PCh. 16 - Prob. 17PCh. 16 - Prob. 18PCh. 16 - Prob. 19PCh. 16 - Prob. 20PCh. 16 - Prob. 21PCh. 16 - Prob. 22PCh. 16 - Prob. 23PCh. 16 - Prob. 24PCh. 16 - Prob. 25PCh. 16 - Prob. 26PCh. 16 - Prob. 27PCh. 16 - Prob. 28PCh. 16 - The mass of a hot-air balloon and its cargo (not...Ch. 16 - Prob. 30PCh. 16 - A popular brand of cola contains 6.50 g of carbon...Ch. 16 - Prob. 32PCh. 16 - At 25.0 m below the surface of the sea, where the...Ch. 16 - To measure how far below the ocean surface a bird...Ch. 16 - Prob. 35PCh. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - A cylinder contains a mixture of helium and argon...Ch. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - (a) How many atoms of helium gas fill a spherical...Ch. 16 - Fifteen identical particles have various speeds:...Ch. 16 - From the MaxwellBoltzmann speed distribution, show...Ch. 16 - Prob. 47PCh. 16 - Helium gas is in thermal equilibrium with liquid...Ch. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - Prob. 52PCh. 16 - A mercury thermometer is constructed as shown in...Ch. 16 - A liquid with a coefficient of volume expansion ...Ch. 16 - A clock with a brass pendulum has a period of...Ch. 16 - A vertical cylinder of cross-sectional area A is...Ch. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - The rectangular plate shown in Figure P16.60 has...Ch. 16 - In a chemical processing plant, a reaction chamber...Ch. 16 - Prob. 62PCh. 16 - Prob. 63PCh. 16 - Two concrete spans that form a bridge of length L...Ch. 16 - A 1.00-km steel railroad rail is fastened securely...Ch. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - Consider an object with any one of the shapes...Ch. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - A cylinder that has a 40.0-cm radius and is 50.0...Ch. 16 - Prob. 75P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- At what temperature is the average speed of carbon dioxide molecules ( M=44.0 g/mol) 510 m/s?arrow_forwardthe copper rod at any temperature. a(steel) (copper) 3. Two glass bulbs of volume 500 and 100 cc are connected by a narrow tube whose volume is negligible. When the apparatus is sealed off, the pressure of the air inside is 70 cm of Hg and the temperature 20°C. What does the pressure become if the 100 cc bulb is kept at 20°C and the other is heated to 100°C?arrow_forwardA Goodyear blimp typically contains 5300 m of helium (He) at an absolute pressure of 1.10 x 10 Pa. The temperature of the helium is 275 K. What is the mass (in kg) of the helium in the blimp? Number Units the tolerance is +/-2%arrow_forward
- Consider a mercury-in-glass thermometer. The capillary tube has a diameter of 0.004 cm, and the bulb has a diameter of 0.25 cm. Suppose that the mercury just fills the bulb at 0.00°C. Neglecting the expansion of the capillary tube so that its cross sectional area is constant, find the change in height of the mercury column if the temperature drops from 23°C to 9°C.arrow_forwardA company advertises that it delivers helium at a gauge pressure of 1.72 × 10 7 Pa in a cylinder of volume 43.8 L. How many balloons can be inflated to a volume of 4.00 L with that amount of helium? Assume the pressure inside the balloons is 1.01 × 105 Pa and the temperature in the cylinder and the balloons is 25.0 °C .arrow_forwardA beachcomber finds a corked bottle containing a message. The air in the bottle is at atmospheric pressure and a temperature of 30.3°C. The cork has a cross-sectional area of 2.10 cm2. The beachcomber places the bottle over a fire, figuring that the increased pressure will push out the cork. At a temperature of 99°C the cork is ejected from the bottle. (a) What was the pressure in the bottle just before the cork left it?arrow_forward
- A sealed container with a lid of area 0.004 m2is filled with an ideal gas. The container and gas are allowed to reach thermal equilibrium with the surrounding air. If a 2000 N block is needed to keep the lid from being pushed off the container, what is the absolute pressure inside the container (the pressure compared to vacuum) ?arrow_forwardAn ideal gas in a 0.15 m cubed container has a pressure of 1.50 times 10 to the 4th power N/m squared at 35.0 degrees Celsius. Determine the number of moles of gas in the container.arrow_forwardThe pressure in a constant-volume gas thermometer is 0.700 atm at 100°C and 0.512 atm at 0°C. (a) What is the temperature when the pressure is 0.0611 atm? °C (b) What is the pressure at 342°C? atmarrow_forward
- A car tire has an inner radius of 45.46 cm has a low pressure of 195 kPa. The tire is recommended to be inflated to 248 kPa. At the low pressure the tire outer radius is 56.65 cm and fully inflated is 67.81 cm. The outside temperature is 17 0C. What is the mass of the air to inflate the tire? How many air molecules are added to properly inflate the tire?arrow_forwardLiquid glycerin is flowing through a 25-mm-diameter and 10-m-long tube. The liquid glycerin enters the tube at 20°C with a mass flow rate of 0.5 kg/s. If the outlet mean temperature is 40°C and the tube surface temperature is constant, determine the surface temperature of the tube.arrow_forwardLiquid water enters a 10-m-long smooth rectangular tube with a = 50 mm and b = 25 mm. The surface temperature is maintained constant, and water enters the tube at 20°C with a mass flow rate of 0.25 kg/s. Determine the tube surface temperature necessary to heat the water to the desired outlet temperature of 80°C.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY