
Soaps, Detergents, and Hard Water
Fatty acid molecules can align to form a barrier called a bilipid layer, shown below. In this schematic, the ionic end of the fatty acid is shown as a circle, and the nonpolar chain is shown as a squiggly line. Use this diagram far questions 66 and 67.
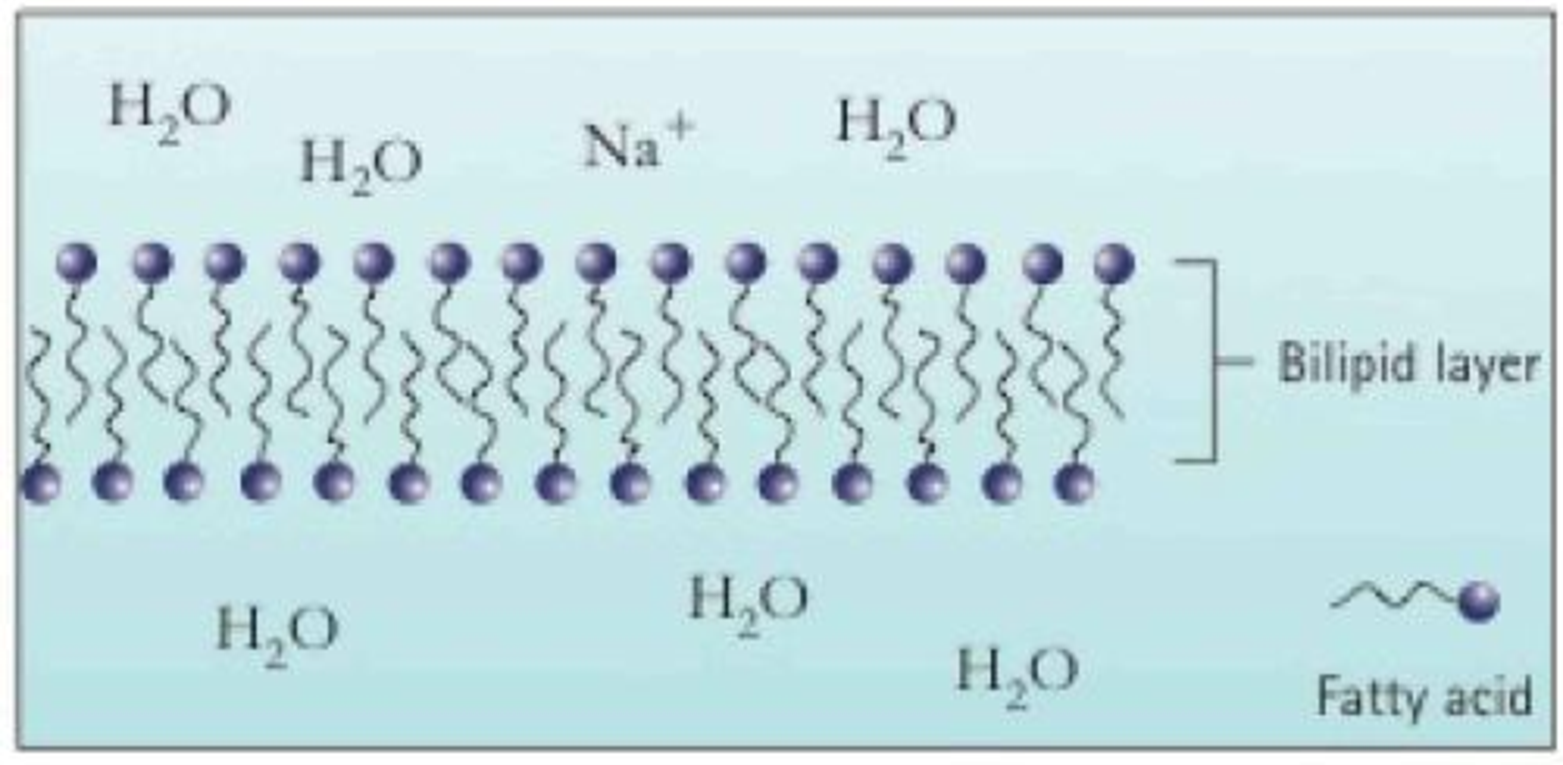
66. Why do nonpolar molecules have a difficult time passing through the bilipid layer? How about polar molecules?
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Conceptual Physical Science, Books a la Carte Edition; Modified Mastering Physics with Pearson eText -- ValuePack Access Card -- for Conceptual Physical Science (6th Edition)
Additional Science Textbook Solutions
Sears And Zemansky's University Physics With Modern Physics
Conceptual Physics (12th Edition)
University Physics Volume 2
The Cosmic Perspective (8th Edition)
Modern Physics
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
- Your roommate is having trouble understanding why solids form. He asks, "Why would atoms bond into solids rather than just floating freely with respect to each other?" To help him understand at least one type of bonding in solids, you decide to embark on an energy explanation. You show him a drawing of a primitive cell of a sodium chloride crystal, NaCl, or simple table salt. The drawing is shown in the figure below, where the orange spheres are Na+ ions and the blue spheres are Cl− ions. Each ion has a charge of magnitude equal to the elementary charge e. The ions lie on the corners of a cube of side d. You explain to your roommate that the electrical potential energy is defined as zero when all eight charges are infinitely far apart from each other. Then you bring them together to form the crystal structure shown. a) Evaluate the electric potential energy of the crystal as shown.arrow_forwardThe surface of an ideal molecule of table salt (NaCl) is a square with a chemical structure as shown in the figure. The blue spheres represent Na+ ions and the green spheres represent Cl− ions. Consider only points A, B, C, and D. What is the magnitude of the net electrostatic force on the chlorine ion at point A due to the sodium ions at points B and C? Note: the bond length is 0.282 nm. (N)arrow_forwardwhat is the physical significance of half adder?arrow_forward
- Shape at she center of, it of afall or mole es Te n heals of a morn is 1 u and that of the Cl atom is 35 u. (b) The H20 molecule is also shown below. The mass of the O atom is 16 u. The H and O atoms are separated by 1 × 10-10 m and the bond angle is 105". Some part A is wrong can you please show all workarrow_forwardA molecule of mass 89.3 atomic mass units moving at 0.80 km/s to the right, and another molecule of mass 18.0 atomic mass units moving at 0.40 km/s to the left, collide. The second molecule has a velocity of 0.60 km/s to the right after the collision. What is the velocity of the first molecule in km/s after the collision? (Don't be thrown by the unusual mass unit here; if you need to just pretend it's grams or kg etc.)arrow_forwardWhy does a carbonated beverage container sizzle upon opening? Why does the beverage taste flat after a period of time?arrow_forward
- Use the data in Table 11.2 to calculate the reduced mass of the NO molecule; then compute a value for using Equation 11.3. Compare the two results.arrow_forwardOne mole of hydrogen peroxide, H2O2, would consist of how many molecules? (13.5) (a) 6.02 1023 (b) 1 (c) 34.0 6.02 1023 (d) 34.0arrow_forwardTwo H2 molecules can react with one O2 molecule to produce two H2O molecules. How many moles of hydrogen molecules ale needed to react with one mole of oxygen molecules?arrow_forward
- Calculate the separation between the two lowest translational energylevels for an O2 molecule in a cubic box with sides of length 5.0 cm. At whatvalue of n = n1 = n2 = n3 does the energy of the molecule reach 32 kT at 300 K,and what is the separation of this level from one of the degenerate levelsimmediately below?please answer type in word don't image upload thank you.arrow_forwardDo covalent compounds conduct electricity when they are mixed with water? Explain.arrow_forwardshow the step by step solution.arrow_forward

 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning





