
Concept explainers
Identify and label the species in each reaction.
(a)
(b)
(a)
Interpretation:
The species in each of the given reaction has to be identified and labeled.
Concept Information:
When a Bronsted acid donates a proton, what remains of the acid is known as a conjugate base; when a Bronsted base accepts a proton, the newly formed protonated species is known as a conjugate acid. This can be given by the below equation as shown in Figure 1.
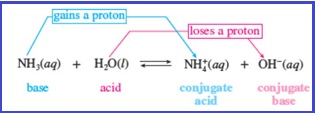
Figure 1
The reactant that loses a proton is the acid and the reactant that gains a proton is the base.
Each product is the conjugate of one of the reactant.
Two species that differ only by a proton constitute a conjugate pair
Addition of proton to a species gives its conjugate acid whereas removal of proton from a species gives its conjugate base.
To Label: The equation
Answer to Problem 2PPA
Answer
Explanation of Solution
Hence,
Therefore, the given equation is labeled as,
(b)
Interpretation:
The species in each of the given reaction has to be identified and labeled.
Concept Information:
When a Bronsted acid donates a proton, what remains of the acid is known as a conjugate base; when a Bronsted base accepts a proton, the newly formed protonated species is known as a conjugate acid. This can be given by the below equation as shown in Figure 1.

Figure 1
The reactant that loses a proton is the acid and the reactant that gains a proton is the base.
Each product is the conjugate of one of the reactant.
Two species that differ only by a proton constitute a conjugate pair
Addition of proton to a species gives its conjugate acid whereas removal of proton from a species gives its conjugate base.
To Label: The equation
Answer to Problem 2PPA
Answer
Explanation of Solution
Hence,
Therefore, the given equation is labeled as,
Want to see more full solutions like this?
Chapter 16 Solutions
Student Solutions Manual For Chemistry: Atoms First
- Define or illustrate the meaning of the following terms: a. Ka reaction b. Ka equilibrium constant c. Kb reaction d. Kb equilibrium constant e. conjugate acidbase pairarrow_forwardWhich of the following is a strong base? a. CH3NH2 b. Ba(OH)2c. Cr(OH)3d. CH3COOHarrow_forward3. Which of the following is an acid and conjugate base pair? a. HCl/OCl- b. H2SO4/SO42- c. NH4+/NH3 d. H3O+/OH-arrow_forward
- Consider the reaction shown below: HCO3-1 (aq) + OH-1 (aq) ⇋⇋ CO3-2(aq) + H2O (l) Identify the Brønsted-Lowry acid in this reaction. Select one: a. HCO3-1 b. OH-1 c. CO3-2 d. H2Oarrow_forwardAs we will see in later chapters, many steps in key reaction sequences involve acid–base reactions. (a) Draw curved arrows to illustrate the flow of electrons in steps [1]–[3]. (b) Identify the base and its conjugate acid in step [1]. (c) Identify the acid and its conjugate base in step [3].arrow_forwardAnswer the following questions about acid-base equilibrium reactions (d)Based on the following Kavalues, which is the strongest acid and which is the weakest acid? Ka (HNO2) = 4.5 x 10 –4Ka (H2SO3) = 1.5 x 10 –2Ka (H2CO3) = 4.3 x 10 –7arrow_forward
- 4. What is the conjugate acid of HCO3^-1arrow_forwardYou may need Table 13-2 to answer the following questions. a. Which is the stronger base, Cl or H2O? b. Which is the stronger base, H2O or NO2? c. Which is the stronger base, CNor OC6H5?arrow_forward. Acetate ion, C2H3O2- , has a stronger affinity for protons than does water. Therefore, when dissolved in water, acetate ion behaves as a(n) _________.arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




