
Concept explainers
Identify and label the species in each reaction.
(a)
(b)
(a)
Interpretation:
The species in each of the given reaction has to be identified and labeled.
Concept Information:
When a Bronsted acid donates a proton, what remains of the acid is known as a conjugate base; when a Bronsted base accepts a proton, the newly formed protonated species is known as a conjugate acid. This can be given by the below equation as shown in Figure 1.
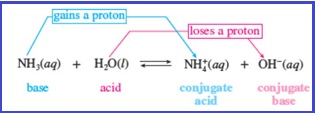
Figure 1
The reactant that loses a proton is the acid and the reactant that gains a proton is the base.
Each product is the conjugate of one of the reactant.
Two species that differ only by a proton constitute a conjugate pair
Addition of proton to a species gives its conjugate acid whereas removal of proton from a species gives its conjugate base.
To Label: The equation
Answer to Problem 2PPA
Answer
Explanation of Solution
Hence,
Therefore, the given equation is labeled as,
(b)
Interpretation:
The species in each of the given reaction has to be identified and labeled.
Concept Information:
When a Bronsted acid donates a proton, what remains of the acid is known as a conjugate base; when a Bronsted base accepts a proton, the newly formed protonated species is known as a conjugate acid. This can be given by the below equation as shown in Figure 1.

Figure 1
The reactant that loses a proton is the acid and the reactant that gains a proton is the base.
Each product is the conjugate of one of the reactant.
Two species that differ only by a proton constitute a conjugate pair
Addition of proton to a species gives its conjugate acid whereas removal of proton from a species gives its conjugate base.
To Label: The equation
Answer to Problem 2PPA
Answer
Explanation of Solution
Hence,
Therefore, the given equation is labeled as,
Want to see more full solutions like this?
Chapter 16 Solutions
CHEM: ATOM FIRST V.2 W/ACCESS >C<
- Define or illustrate the meaning of the following terms: a. Ka reaction b. Ka equilibrium constant c. Kb reaction d. Kb equilibrium constant e. conjugate acidbase pairarrow_forwardWhich of the following is a strong base? a. CH3NH2 b. Ba(OH)2c. Cr(OH)3d. CH3COOHarrow_forward3. Which of the following is an acid and conjugate base pair? a. HCl/OCl- b. H2SO4/SO42- c. NH4+/NH3 d. H3O+/OH-arrow_forward
- Consider the reaction shown below: HCO3-1 (aq) + OH-1 (aq) ⇋⇋ CO3-2(aq) + H2O (l) Identify the Brønsted-Lowry acid in this reaction. Select one: a. HCO3-1 b. OH-1 c. CO3-2 d. H2Oarrow_forwardAs we will see in later chapters, many steps in key reaction sequences involve acid–base reactions. (a) Draw curved arrows to illustrate the flow of electrons in steps [1]–[3]. (b) Identify the base and its conjugate acid in step [1]. (c) Identify the acid and its conjugate base in step [3].arrow_forwardAnswer the following questions about acid-base equilibrium reactions (d)Based on the following Kavalues, which is the strongest acid and which is the weakest acid? Ka (HNO2) = 4.5 x 10 –4Ka (H2SO3) = 1.5 x 10 –2Ka (H2CO3) = 4.3 x 10 –7arrow_forward
- 4. What is the conjugate acid of HCO3^-1arrow_forwardYou may need Table 13-2 to answer the following questions. a. Which is the stronger base, Cl or H2O? b. Which is the stronger base, H2O or NO2? c. Which is the stronger base, CNor OC6H5?arrow_forward. Acetate ion, C2H3O2- , has a stronger affinity for protons than does water. Therefore, when dissolved in water, acetate ion behaves as a(n) _________.arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




