
Chemistry: Structure And Properties: Custom Edition For University Of Maryland, College Park
4th Edition
ISBN: 9781323738832
Author: Tro
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 17, Problem 147E
Interpretation Introduction
To determine:
If p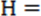 p
p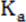 , p
, p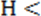 p
p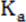 , or p
, or p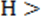 p
p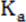 .
.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1) In the laboratory, a general chemistry student measured the pH of a 0.554 M aqueous solution of nitrous acid to be 1.787.
Use this information she obtained to determine the Ka for this acid?
Ka .......................... ?
2) How many grams of solid potassium hydroxide ( molar mass= 56.1 g/mol) are needed to react with 26.7 mL of a 1.75 M hydroiodic acid solution?
KOH + HI......> kI + H2o
................ grams
Without doing any calculations, determine if pH = pKa, pH 7 pKa, or pH 6 pKa. Assume that HA is a weak monoprotic acid.a. 0.10 mol HA and 0.050 mol of A- in 1.0 L of solutionb. 0.10 mol HA and 0.150 mol of A- in 1.0 L of solutionc. 0.10 mol HA and 0.050 mol of OH- in 1.0 L of solutiond. 0.10 mol HA and 0.075 mol of OH- in 1.0 L of solution
The ability of a solution to resist changes to the pH from added acid or base depends upon the amount of weak acid and weak base. The buffering capacity is the ratio of weak acid to weak base and the best buffering occurs when these are equal, making the buffering capacity equal to 1. In the above buffer the buffering capacity is the ratio of
[H2PO4-1] to [HPO4-2], or
Ka [H2PO4-1] Ka (4.44 x 10-5) Ka (8)
[H3O+1] = ––––––––––––– = –––––––––––––––– = –––––––––
[HPO4-2] (5.55 x 10-5) (10)
Here the buffering capacity is close to 1 (0.8) and so the buffering capacity is Blank 1. Fill in the blank, read surrounding text.
When the buffering capacity is 1, the [H3O+1] = Ka
and the pH = pKa
This helps chemists choose the right buffer for the needed pH. To buffer at pH = 7,
the weak acid should a pKa of close to 7, True or False ? Blank 2. Fill in the blank, read surrounding text.
the weak…
Chapter 17 Solutions
Chemistry: Structure And Properties: Custom Edition For University Of Maryland, College Park
Ch. 17 - What is the pH range of human blood? How is human...Ch. 17 - What is a buffer? How does a buffer work? How does...Ch. 17 - What is the common ion effect?Ch. 17 - What is the HendersonHasselbalch equation, and why...Ch. 17 - What is the pH of a buffer when the concentrations...Ch. 17 - Suppose that a buffer contains equal amounts of a...Ch. 17 - How do you use the Henderson—Hasselbalch equation...Ch. 17 - What factors influence the effectiveness of a...Ch. 17 - What is the effective pH range of a buffer...Ch. 17 - Describe acidbase titration. What is the...
Ch. 17 - The pH at the equivalence point of the titration...Ch. 17 - The volume required to reach the equivalence point...Ch. 17 - In the titration of a strong acid with a strong...Ch. 17 - In the titration of a weak acid with a strong...Ch. 17 - The titration of a diprotic acid with sufficiently...Ch. 17 - In the titration of a polyprotic acid, the volume...Ch. 17 - What is the difference between the endpoint and...Ch. 17 - What is an indicator? How can an indicator signal...Ch. 17 - What is the solubility-product constant? Write a...Ch. 17 - What is molar solubility? How do you obtain the...Ch. 17 - How does a common ion affect the solubility of a...Ch. 17 - How is the solubility of an ionic compound with a...Ch. 17 - For a given solution containing an ionic compound,...Ch. 17 - What is selective precipitation? Under which...Ch. 17 - In which of these solutions does HNO2 ionize less...Ch. 17 - A formic acid solution has a pH of 3.25. Which of...Ch. 17 - Solve an equilibrium problem (using an ICE table)...Ch. 17 - Solve an equilibrium problem (using an ICE table)...Ch. 17 - Calculate the percent ionization of a 0.15 M...Ch. 17 - Calculate the percent ionization of a 0.13 M...Ch. 17 - Solve an equilibrium problem (using an ICE table)...Ch. 17 - Solve an equilibrium problem (using an ICE table)...Ch. 17 - A buffer contains significant amounts of acetic...Ch. 17 - A buffer contains significant amounts of ammonia...Ch. 17 - Use the HendersonHasselbalch equation to calculate...Ch. 17 - Use the Henderson—Hasselbalch equation to...Ch. 17 - Use the Henderson—Hasselbalch equation to...Ch. 17 - Use the Henderson—Hasselbaich equation to...Ch. 17 - Calculate the pH of the solution that results from...Ch. 17 - Calculate the pH of the solution that results from...Ch. 17 - Calculate the ratio of NaF to HF required to...Ch. 17 - Calculate the ratio of CH3NH2 to CH3NH3Cl...Ch. 17 - What mass of sodium benzoate should you add to...Ch. 17 - What mass of ammonium chloride should you add to...Ch. 17 - A 250.0-mL buffer solution is 0.250 M in acetic...Ch. 17 - A 100.0-mL buffer solution is 0.175 M in HCIO and...Ch. 17 - For each solution, calculate the initial and final...Ch. 17 - For each solution, calculate the initial and final...Ch. 17 - A 350.0-mL buffer solution is 0.150 in HF and...Ch. 17 - A 100.0-mL buffer solution is 0.100 M ¡n NH3 and...Ch. 17 - Determine whether the mixing of each pair of...Ch. 17 - Determine whether the mixing of each pair of...Ch. 17 - Blood s buffered by carbonic acid and the...Ch. 17 - The fluids within cells are buffered by H2PO4 and...Ch. 17 - Which buffer system is the best choice to create a...Ch. 17 - Which buffer system is the best choice to create a...Ch. 17 - A 500.0-mL buffer solution is 0.100 M in HNO2 and...Ch. 17 - Prob. 58ECh. 17 - The graphs labeled (a) and (b) are the titration...Ch. 17 - Two 25.0-mL samples, one 0.100 M HCI and the other...Ch. 17 - Two 20.0-mL samples, one 0.200 M KOH and the other...Ch. 17 - Prob. 62ECh. 17 - Consider the curve shown here for the titration of...Ch. 17 - Consider the curve shown here for the titration of...Ch. 17 - Consider the titration of a 35.0-mL sample of...Ch. 17 - A 20.0-mL sample of 0.125 M HNO3 is titrated with...Ch. 17 - Consider the titration of a 25.0-mL sample of...Ch. 17 - Prob. 68ECh. 17 - Prob. 69ECh. 17 - Prob. 70ECh. 17 - Consider the titration of a 25.0-mL sample of...Ch. 17 - Prob. 72ECh. 17 - Prob. 73ECh. 17 - Prob. 74ECh. 17 - Prob. 75ECh. 17 - Prob. 76ECh. 17 - Prob. 77ECh. 17 - Prob. 78ECh. 17 - Methyl red has a pKaof 5.0 and is red in its acid...Ch. 17 - Phenolphthalein has a pKaof 9.7. It is colorless...Ch. 17 - Referring to Table 17.1pick an indicator for use...Ch. 17 - Referring to Table 17.1 pick an indicator for use...Ch. 17 - Write balanced equations and expressions for...Ch. 17 - Prob. 84ECh. 17 - Refer to the Kspvalues in Table 17.2 to calculate...Ch. 17 - Prob. 86ECh. 17 - Use the given molar solubilities in pure water to...Ch. 17 - Prob. 88ECh. 17 - Two compounds with general formulas AX and AX2...Ch. 17 - Consider the compounds with the generic formulas...Ch. 17 - Refer to the Ksp value from Table 17.2 to...Ch. 17 - Prob. 92ECh. 17 - Calculate the molar solubility of barium fluoride...Ch. 17 - Prob. 94ECh. 17 - Calculate the molar solubility of calcium...Ch. 17 - Calculate the solubility (in grams per 1.00102 of...Ch. 17 - Is each compound more soluble in acidic solution...Ch. 17 - Is each compound more soluble in acidic solution...Ch. 17 - A solution containing sodium fluoride is mixed...Ch. 17 - A solution containing potassium bromide is mixed...Ch. 17 - Predict whether a precipitate forms if you mix...Ch. 17 - Prob. 102ECh. 17 - Prob. 103ECh. 17 - Prob. 104ECh. 17 - A solution is 0.010 M in Ba2+ and 0.020 M in Ca2+...Ch. 17 - Prob. 106ECh. 17 - A solution is made 1.1103M in Zn(NO3)2 and 0.150 M...Ch. 17 - A 120.0-mL sample of a solution that is 2.8103M in...Ch. 17 - Use the appropriate values of Kspand Kfto find the...Ch. 17 - Prob. 110ECh. 17 - A 1.500-mL solution contains 2.05 g of sodium...Ch. 17 - A solution ¡s made by combining 10.0 ml of 17.5 M...Ch. 17 - A buffer is created by combining 150.0 mL of 0.25...Ch. 17 - A buffer is created by combining 3.55 g of NH3...Ch. 17 - A 1.0-L buffer solution initially contains 0.25...Ch. 17 - A 250.0-mL buffer solution initially contains...Ch. 17 - In analytical chemistry, bases used for titrations...Ch. 17 - A 0.5224-g sample of an unknown monoprotic acid...Ch. 17 - A 0.25-mol sample of a weak acid with an unknown...Ch. 17 - A 5.55-g sample of a weak acid with Ka=1.3104 is...Ch. 17 - A 0.552-g sample of ascorbic acid (vitamin C) is...Ch. 17 - Sketch the titration curve from Problem 121by...Ch. 17 - One of the main components of hard water is CaCO3....Ch. 17 - Gout—a condition that results in joint swelling...Ch. 17 - Pseudogout, a condition with symptoms similar to...Ch. 17 - Calculate the solubility of silver chloride in a...Ch. 17 - Calculate the solubility of CuX ¡n a solution that...Ch. 17 - Aniline, C6H5NH2, is an important organic base...Ch. 17 - The Kbof hydroxylamine, NH2OH is 1.0108 . A buffer...Ch. 17 - Prob. 130ECh. 17 - Prob. 131ECh. 17 - Prob. 132ECh. 17 - What relative masses of dimethyl amine and...Ch. 17 - You are asked to prepare 2.0 L of a HCN/NaCN...Ch. 17 - Prob. 135ECh. 17 - Prob. 136ECh. 17 - Prob. 137ECh. 17 - Prob. 138ECh. 17 - When excess solid Mg(OH)2 is shaken with 1.00 L of...Ch. 17 - Prob. 140ECh. 17 - Calculate the solubility of Au(OH)3 in (a) water...Ch. 17 - Calculate the concentration of I in a solution...Ch. 17 - Prob. 143ECh. 17 - Prob. 144ECh. 17 - Find the pH of a solution prepared from 1.0 L of a...Ch. 17 - Prob. 146ECh. 17 - Prob. 147ECh. 17 - Prob. 148ECh. 17 - Consider three solutions: 0.10 M solution of a...Ch. 17 - Prob. 150ECh. 17 - Prob. 151ECh. 17 - Prob. 152ECh. 17 - Prob. 153ECh. 17 - Prob. 154ECh. 17 - A certain town gets its water from an underground...Ch. 17 - Prob. 156ECh. 17 - Prob. 157ECh. 17 - A buffer is 0.100 M in NH4CI and 0.100 M in NH3....Ch. 17 - What is the pH of a buffer that is 0.120 M in...Ch. 17 - Prob. 3SAQCh. 17 - Prob. 4SAQCh. 17 - Prob. 5SAQCh. 17 - Prob. 6SAQCh. 17 - Prob. 7SAQCh. 17 - A 10.0-mL sample of 0.200 M hydrocyanic acid (HCN)...Ch. 17 - Prob. 9SAQCh. 17 - Prob. 10SAQCh. 17 - Prob. 11SAQCh. 17 - Prob. 12SAQCh. 17 - Calculate the molar solubility of magnesium...Ch. 17 - Prob. 14SAQCh. 17 - Prob. 15SAQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- HCN (aq) + H2O (l) ⇄ H3O+ (aq) + CN- (aq) Ka = ([H3O+][CN-])/[HCN] = 6.2 × 10-10 The equilibrium reaction shown above represents the partial ionization of the weak acid HCN (aq). A 0.200 M HCN (aq) solution has a pH ≈ 4.95. If 0.05 g (0.010 mol) of NaCN (s) is added to 100 mL of 0.200 M HCN (aq), which of the following explains how and why the pH of the solution changes? The pH will be higher than 4.95 because adding CN- will disrupt the equilibrium, resulting in an increased production of HCN that decreases the concentration of H3O+. The pH will be lower than 4.95 because adding CN- will disrupt the equilibrium, resulting in an increased production of HCN that decreases the concentration of H3O+. The pH will be higher than 4.95 because CN- is a strong base that can neutralize HCN. The pH will remain close to 4.95 because the Ka is so small that hardly any products form.arrow_forwardHCN (aq) + H2O (l) ⇄ H3O+ (aq) + CN- (aq) Ka = ([H3O+][CN-])/[HCN] = 6.2 × 10-10 The equilibrium reaction shown above represents the partial ionization of the weak acid HCN (aq). A 0.200 M HCN (aq) solution has a pH ≈ 4.95. If 0.05 g (0.010 mol) of NaCN (s) is added to 100 mL of 0.200 M HCN (aq), which of the following explains how and why the pH of the solution changes?arrow_forwardCalculate the pH of each of the following solutions a. 0.10 M propanoic acid, CH3CH2COOH, Ka = 1.3 x 10-5 b. 0.10 M sodium propanoate, NaCH3CH2COO. c. A mixture containing 0.10 M propanoic acid and 0.10 M sodium propanoate d. Calculate the pH after 0.020 mole of HCl is added to 1.00 L of a solution that contains 0.10 M propanoic acid and 0.10 M sodium propanoate e. Calculate the pH after 0.020 mole of NaOH is added to 1.00 L of a solution that contains 0.10 M propanoic acid and 0.10 M sodium propanoatearrow_forward
- The Ka of an acid is known to be 1.51 X 10^-5. A solution is made by adding 0.279 mol of the acid to enough water to make the final volume 1.30 L. a. what is the equilibrium concentration of H_3O+ b. what is the equilibrium pH of the solution c. what is the equilibrium concentration of the conjugate base d. what is the equilibruim concentration of the acid e. what is the % dissociation of the acidarrow_forwardCalculate the pH of the resulting solution when 100.0 mL of 0.2 M HNO2 is titrated by 0.1 M NaOH after d) 200 mL of NaOH have been added. e) 300 mL of NaOH have been added. The Ka of HNO2 is 4.5 x 10-4arrow_forwardHow many milliliters of 0.0200 M Ca(OH)₂are required to neutralize 79.7 mL of 0.0300 M HCl? Calculate the pH when 80.5 mL of 0.200 M HBr is mixed with 30.0 mL of 0.400 M CH₃NH₂ (Kb = 4.4 × 10⁻⁴).arrow_forward
- 100.0 ml of a 0.3 M solution of an unknown monoprotic weak acid has a Ka=1x10-6. What mass of NaOH needs to be added to the solution to achieve a pH of 5.1?arrow_forward1. Consider the titration of a 60.0 mL of 0.347 M weak acid HA (Ka = 4.2 x 10⁻⁶) with 0.400 M KOH. What is the pH before any base has been added? 2. Consider the titration of a 60.0 mL of 0.347 M weak acid HA (Ka = 4.2 x 10⁻⁶) with 0.400 M KOH. After 30.0 mL of KOH have been added, what would the pH of the solution be? 3.Consider the titration of a 60.0 mL of 0.347 M weak acid HA (Ka = 4.2 x 10⁻⁶) with 0.400 M KOH. After 75.0 mL of KOH have been added, what would the pH of the solution be?arrow_forwardWeak Acids dissociate according to the following equation: HA(aq) + H2O(L) ⇌ H3O+ (aq) + A-(aq) In a lab experiment, a student needs to make a buffer solution with a pH of 6.870. If the pKa of the weak acid is 6.500, what ratio of conjugate weak base to acid is needed to make the buffer, I.e. what is the value of [A-/HA]? (Please provide your answer to 3 decimal places.)arrow_forward
- Calculate the change in pH when 9.00 of 0.100 M HCl * (aq) is added to 100.0 of a buffer solution that is 0.100 M NH 3 (aq) and 0.100 M in NH 4 Cl(aq) . Consult the table of ionization constants as needed. Delta*p * H = Calculate the change in pH when 9.00 mL of 0.100 M NaOH is added to the original buffer solution. Delta*p * H =arrow_forwardthe answer I got for this question is wrong, along with quite a few I have asked in the past. The Ph of a weak acid and strong base is not 7. It does not work that way. Þú byrjar með 100.0 mL af 0.250 M HCN og bætir við 0.750 M NaOH við 25°C. (HCN Ka = 6.2 * 10-10.) You begin with 100 mL of 0,250 M HCN and add to it 0,75 M NaOH at 25°C (HCN Ka = 6,2 * 10-10) Calculate the pH at the equivalence point. / Calculate the pH after 150 mL of NaOH has been added. Can you please check this answer and help me out https://www.bartleby.com/questions-and-answers/you-begin-with-100-ml-of-0250-m-hcn-and-add-to-it-075-m-naoh-at-25c-hcn-ka-62-10-10-calculate-the-ph/9cc945c7-a648-46e2-8057-4c800a5eb855arrow_forwardWhat is the pH of a buffer system prepared by dissolving 12.00 grams of NH4Cl and 40.00mL of 12 M NH3 in enough water to make 1.0L of solution? Kb=1.80*10^-5 for NH3- what is the pH of a solution made by mixing 30 mL of 0.100 M HCl with 40.0 mL of 0.100 M KOH? Assume that the total volume is 70mLarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY