
Interpretation:
The functional groups present in lidocaine has to be identified. Also the structure of carboxylic acid and
Concept Introduction:
Carboxylic acid: One
Amide: One
Amide Formation: Amide is formed when a carboxylic acid reacts with an amine or ammonia.
- Primary amide is produce when a carboxylic acid reacts with ammonia.
- Secondary and tertiary amide is produce when a carboxylic acid reacts with primary and secondary amine respectively.
- Ammonium salt is formed when tertiary amine and a carboxylic acid reacts forming an ionic compound with a carboxylate acid anion and a trialkyl ammonium cation since there is no hydrogen atom in trialkyl amine
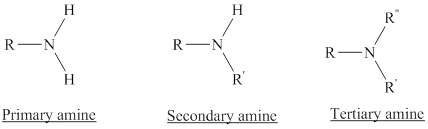
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Aromatic Compounds: Compounds that are planar, cyclic and having
Trending nowThis is a popular solution!

Chapter 17 Solutions
FUND. GEN. ORG&BIO CHEM PKG
- List two classes of compounds derived from arachidonic acid. Suggest some reasons for the amount of biomedical research devoted to these compounds.arrow_forwardFrom which name reaction Lorazepam can be synthesized?arrow_forwardHow can chirality and stereoisomers influence the pharmacology, bioactivity, toxicology, pharmacokinetics, and metabolism of ibuprofen? Please provide a detailed summary of what might happen if, for instance, the R enantiomer isn't able to be inverted to the bioactive S enantiomer.arrow_forward
- What would be the characteristics of a transition-state analog for the chymotrypsin reaction?arrow_forwardWhy is salbutamol better than isoproterenol in asthma management in terms of their chemical structure?arrow_forwardName at least two undesirable characteristics are often associated with alkaloids.arrow_forward
- How aspirin is prepared from salicylic acid?arrow_forwardExplain in terms of the Michaelis-Menten equation why alcohol degradation is not first-orderwhile most other drugs are.arrow_forwardHow do N-acetyl- D-glucosamine and N-acetyl- D-galactosamine differ in structure? Are these two compounds constitutional isomers or stereoisomers? Explain your choicearrow_forward
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
