
Concept explainers
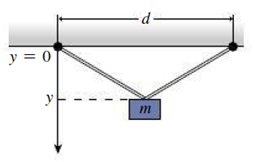
Figure 17.12 shows an apparatus used to determine the linear expansion coefficient of a metal wire. The wire is attached to two points a distance d apart (you don’t know d). A mass hangs from the middle of the wire. The wire’s total length is 100.00 cm at 0°C. The distance y from the suspension points to the top of the mass is measured, and the results are given in the table below. (a) Find an expression for v as a function of temperature, and manipulate your expression to get a linear relation between some function of y and some function of temperature T. You’ll encounter the expression L2 where L is the length of the wire, and, because the change in length is small, you can drop terms involving α2 when you expand L2. (b) Calculate the quantities in your relation from the given data, and plot. Determine a best-fit line and use it to determine the coefficient of linear expansion a and the separation d. (c) Consult Table 17.2 to identify the metal the wire is made of. Ignore any stretching of the wire due to its “springiness”; that is, consider only thermal expansion.
| Temperature. T (°C) | 0 | 20 | 40 | 60 | 80 | 100 | 120 |
| y (cm) | 30.00 | 30.05 | 30.07 | 30.11 | 30.16 | 30.19 | 30.24 |
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Essential University Physics (3rd Edition)
Additional Science Textbook Solutions
Essential University Physics: Volume 2 (3rd Edition)
University Physics Volume 1
College Physics
Conceptual Physical Science (6th Edition)
Cosmic Perspective Fundamentals
Physics for Scientists and Engineers with Modern Physics
- On a hot summer day, the density of air at atmospheric pressure at 35.0C is 1.1455 kg/m3. a. What is the number of moles contained in 1.00 m3 of an ideal gas at this temperature and pressure? b. Avogadros number of air molecules has a mass of 2.85 102 kg. What is the mass of 1.00 m3 of air? c. Does the value calculated in part (b) agree with the stated density of air at this temperature?arrow_forwardConsider the figure attached. An illustration of a circular steel casting with a gap. (a) If the casting is heated, does the width of the gap increase or decrease? a. increase b. decrease (b) The gap width is 1.510 cm when the temperature is 30.0°C. Determine the gap width when the temperature is 175°C. (Use 11 ✕ 10−6 (°C)−1 for the average coefficient of linear expansion for steel. Give your answer to at least three decimal places.)______ cmarrow_forwardThe area A of a rectangular plate is ab = 1.4 m². Its coefficient of linear expansion is a = 22 × 10° /C°. After a temperature rise AT = 17°C, side a is longer by Aa and side b is longer by Ab (see the figure). Neglecting the small quantity (AaAb)/ab, find AA. a Да Aa · Ab Ab Number i Unitsarrow_forward
- Question 4arrow_forwardA circular hole in an aluminum plate is 2.386 cm in diameter at 0.000°C. What is its diameter when the temperature of the plate is raised to 75.50°C? The linear expansion coefficient of aluminum is 23.00 x 10-6 /C°. Number Units Use correct number of significant digits; the tolerance is +/-2%arrow_forwardThe pressure in a constant-volume gas thermometer is 0.700 atm at 100°C and 0.512 atm at 0°C. (a) What is the temperature when the pressure is 0.0611 atm? °C (b) What is the pressure at 342°C? atmarrow_forward
- An aluminum cup of 97 cm³ capacity is completely filled with glycerin at 17°C. How much glycerin will spill out of the cup if the temperature of both the cup and glycerin is increased to 32°C? (The linear expansion coefficient of aluminum is 23 × 10-6 1/C°. The coefficient of volume expansion of glycerin is 5.1 × 10-4 1/C°.) Number i Unitsarrow_forwardThe band in Figure P10.23 is stainless steel (coefficient of linear expansion 5 17.3 × 10–6 °C–1; Young’s modulus 5 18 × 1010 N/m2). It is essentially circular with an initial mean radius of 5.0 mm, a height of 4.0 mm, and a thickness of 0.50 mm. If the band just fits snugly over the tooth when heated to a temperature of 80°C, what is the tension in the band when it cools to a temperature of 37°C?arrow_forwardAn aluminum can is filled to the brim with a liquid. The can and the liquid are heated so their temperatures change by the same amount. The can’s initial volume at 15 oC is 4.5×10- 4 m3 . The coefficient of volume expansion for aluminum is 69×10-6 oC-1 . When the can and the liquid are heated to 75 oC, 2.9×10-6 m3 of liquid spills over. What is the coefficient of volume expansion of the liquid?arrow_forward
- Consider a flat metal plate with width w and length l, so its area is A = lw. The metal has coefficient of linear expansion α. Derive an expression that gives the change ΔA in area for a change ΔT in temperature. Express your answer in terms of the variables α, A, and ΔT for change in temperature.arrow_forwardA construction worker uses a steel tape to measure the length of an aluminum support column. If the measured length is 19.700 m when the temperature is 21.2°C, what is the measured length when the temperature rises to 29.4°C? Note: Don't neglect the expansion of the tape. (Enter your answer to at least three decimal places.)arrow_forwardYou do an experiment in which you transfer energy to 1.2 moles of a gas and measure it’s change in temperature. You make a graph of the data as shown. The y-axis is the amount of energy added and the x-axis is the temperature. A) how many moles per molecule does this substance have? B) is the substance most likely a monatomic gas, diatomic or another type of gas?arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning

