
Concept explainers
(a)
Interpretation: The following compound should be classified according to its
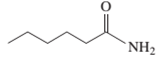
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
(b)
Interpretation: The following compound should be classified according to its functional group:
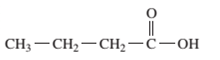
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
(c)
Interpretation: The following compound should be classified according to its functional group:
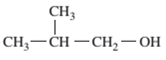
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
(d)
Interpretation: The following compound should be classified according to its functional group:
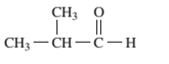
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK BASIC CHEMISTRY
- Among NaHCO3, Vinegar, and Alcohol which among these can be used to remove grease and fats from pans through heatingarrow_forwardIf 60.0 g each of acetic acid (CH3CO2H) and ethanol (C,H5OH) are allowed to react in a 1.00-L sealed container until equilibrium is established, how many moles of the ester (CH3CO2C2H5)and water are formed, and how many moles of ethanol and acid remain? (K = 4.00.) CH3CO2H + C2H5OH CH;CO2C2H5 + H2Oarrow_forwardCERCLA is one of the acts which has established the regulations for hazardous waste. CERCLA relates to: a toxicity of hazardous waste b disposal of hazardous waste c hazardous waste transportation d currently generated hazardous waste e clean up and past practices of hazardous wastearrow_forward
- C7H6O3 + C4H6O3 <-> C9H8O4 + C2H4O2 1.0g of salicylic acid reacts completely with an recess if acetic anhydride. Whats the theoretical yield of aspirin in G?arrow_forwardfive (5) active compounds present in strawberry ( organic chemistry )arrow_forward12.60 Which of the following aldehydes or ketones are soluble in water? (12.3) |3| a. CH3-CH2 -C-CH3 b. CH3 — С —н CH3 c. CH3-CH2-CH-CH2-CH2-C-Harrow_forward
- Q#5. (5 points) Write the chemical equation for the Fisher esterification reaction of acetic acid and isoamyl alcohol and a strong acid catalyst using chemical structures. 4arrow_forwardRank the following compounds number 20 onlyarrow_forwardAlkaloids are basic nitrogen-containing compounds of plant origin, many of which are physiologically active when administered to humans. Ingestion of coniine, isolated from water hemlock, can cause weakness, labored respiration, paralysis, and eventually death. Coniine is the toxic substance in the “poison hemlock” used in the death of Socrates. In small doses, nicotine is an addictive stimulant. In larger doses, it causes depression, nausea, and vomiting. In still larger doses, it is a deadly poison. Solutions of nicotine in water are used as insecticides. Cocaine is a central nervous system stimulant obtained from the leaves of the coca plant.Classify each amino group in these alkaloids according to type (primary, secondary, tertiary, aliphatic, aromatic, heterocyclic).arrow_forward
- CH3 + KMNO4 CH3 CH2 85% H¿PO4 CH3 CH3 Q2. In the Preparation of Alkenes from alcohols the acid (HA) is used as dehydrating agent shoul their ion (A) be (At the same time): 1. 2.arrow_forwardpart: 6b: a,b,carrow_forwardWhich of the following statements is true16. lodoform, which has a molecular formula of CHal, is indicative that a ketomethyl group is present. 17. Based on the physical property, hydroquinone has a lower boiling point compared to catechol. 18. When FeCl3 is used in visualizing aspirin in thin layer chromatography, the resulting spot is red in color.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

