
Concept explainers
(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: 1.
(a)
Interpretation: The IUPAC name for A and B is to be determined.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the carbon chain containing higher number of carbon atoms.
2. For functional group such as aldehyde suffix ‘al’ and for ketone suffix ‘one’ is added to the name.
3. When the chain is substituted with different substituents, then the numbering is done according to priority.
Answer to Problem 37P
The IUPAC name for A and B is
Explanation of Solution
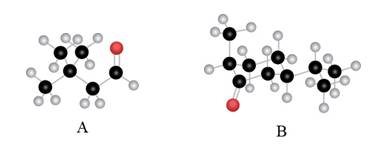
The given compound is,
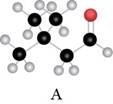
Figure 1
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure is,
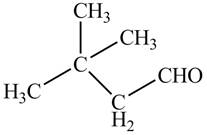
Figure 2
The parent hydrocarbon is butane. The functional group present is aldehyde. Two methyl groups are present on third carbon atom. When same substituents are present, then prefix depends on the number of substituents. The IUPAC name of the compound is
The given compound is,
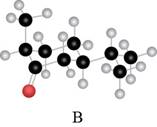
Figure 3
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure is,
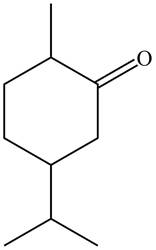
Figure 4
The parent hydrocarbon is cyclohexane. The functional group present is ketone. Methyl group is present on fifth carbon atom and isopropyl group is present on third carbon atom. The IUPAC name of the compound is
The IUPAC name for A and B is
(b)
Interpretation: The products formed when B is treated with given reagents are to be drawn.
Concept introduction: The metal hydride reagents are good reducing agents such as
Grignard reagents are organometallic compounds which are prepared using alkyl halides in presence of magnesium metal in dry ether. These reagents act as strong nucleophiles and bases.
Answer to Problem 37P
The products formed when B is treated with given reagents are,
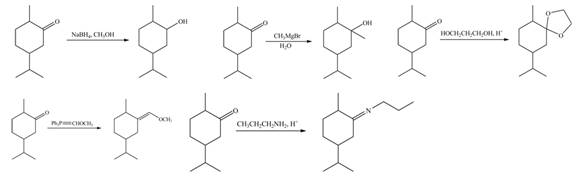
Explanation of Solution
1. In the presence of
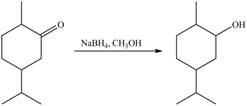
Figure 5
The product formed is
2. The Grignard reagent reacts with compound B to form secondary alcohol. The corresponding reaction is shown below.
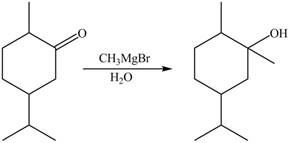
Figure 6
The product formed is
3. When compound B is treated with
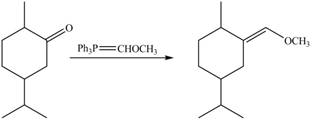
Figure 7
The product formed is
4. The compound B reacts with
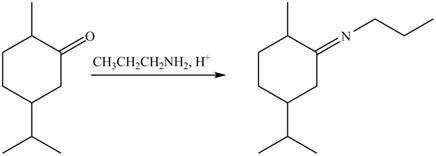
Figure 8
The product formed is
5. The product formed by the reaction of compound B with ethylene glycol in the presence of
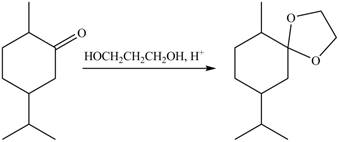
Figure 9
The products formed when B is treated with given reagents are shown in Figure 5, Figure 6, Figure 7, Figure 8 and Figure 9.
Want to see more full solutions like this?
Chapter 18 Solutions
EBK ORGANIC CHEMISTRY
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardSpell out the IUPAC name(s) of the alcohol(s). If there is more than one alcohol, spell out the full names separated by a comma.arrow_forwardDraw the organic products for A and B.arrow_forward
- What products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardThe action of heating one mole of water (containing a trace of acid catalyst) on one mole of compound Y produces one mole of CH,COOH and one mole of CH, CH,NH, The structure of Y is: Select one: O CHCNCCH, CH,CH,NHCH,CH, OH CH CHNHCH,CH, O CHCH,CNHCH; O CH;CH NHÖCH;arrow_forwardGive the IUPAC names of the ff:arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
