
PHYSICS:F/SCI.+ENGRS.,V.1
10th Edition
ISBN: 9781337553575
Author: SERWAY
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 40AP
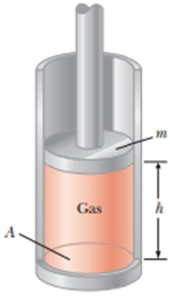
A vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P18.40). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find the height h in Figure P18.40. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder.
Figure P18.40
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Many fish maintain buoyancy with a gas-filled swim bladder. The pressure inside the swim bladder is the same as the outside water pressure, so when a fish descends to a greater depth, the gas compresses. Adding gas to restore the original volume requires energy. A fish at a depth where the absolute pressure is 3.0 atm has a swim bladder with the desired volume of 5.0 × 10-4 m3. The fish now descends to a depth where the absolute pressure is 5.0 atm.a. The gas in the swim bladder is always the same temperatureas the fish’s body. What is the volume of the swim bladder at the greater depth?b. The fish remains at the greater depth, slowly adding gas to the swim bladder to return it to its desired volume. How much work is required?
A frictionless and leak-free piston moves up
and down a cylinder whose diameter and
height are 10 cm and 1 m, respectively (see
the sketch). At equilibrium, the downward
force on the piston, due to the atmospheric
pressure (100 kPa) and the gravitational force
on the mass of the piston, is equal to the
magnitude of the upward force on the piston,
due to the pressure of gas in the cylinder. The
cylinder is filled with NH3 at 150 kPa and 20 °
C, which maintains the piston to its initial
location of H = 80 cm. Assuming that the ideal gas law is applicable -
Pirton
NH,
(a) Calculate the mass of the piston.
(b) Calculate the mass of NH3 in the cylinder. Use, R = 0.4882 kJ/kg.K
%3D
A metal weight with a mass of 40.03 kg is now placed on the piston,
which causes it to move downward in the cylinder.
(c) Calculate the pressure in the cylinder and the piston location (H)
if the temperature of Ammonia remains constant at 20 ° C.
(d) Find the temperature to which the NH3 in the cylinder must be…
A hand-driven tire pump has a piston with a diameter d and a maximum stroke of 30.0 cm. You Find the diameter d of the piston in the above question, If the work done in one stroke is 30.0 J and if the average gauge pressure is 2.40 x 105 N/m2 (a) Find the diameter d of the piston (b) What average force do you exert on the piston, neglecting friction and gravitational force?
Chapter 18 Solutions
PHYSICS:F/SCI.+ENGRS.,V.1
Ch. 18.1 - Prob. 18.1QQCh. 18.3 - Consider the following pairs of materials. Which...Ch. 18.4 - If you are asked to make a very sensitive glass...Ch. 18.4 - Two spheres are made of the same metal and have...Ch. 18.5 - A common material for cushioning objects in...Ch. 18.5 - On a winter day, you turn on your furnace and the...Ch. 18 - Prob. 1PCh. 18 - Prob. 2PCh. 18 - Prob. 3PCh. 18 - Liquid nitrogen has a boiling point of 195.81C at...
Ch. 18 - Death Valley holds the record for the highest...Ch. 18 - Prob. 6PCh. 18 - A copper telephone wire has essentially no sag...Ch. 18 - A pair of eyeglass frames is made of epoxy...Ch. 18 - The Trans-Alaska pipeline is 1 300 km long,...Ch. 18 - A square hole 8.00 cm along each side is cut in a...Ch. 18 - You are watching a new bridge being built near...Ch. 18 - You are watching a new bridge being built near...Ch. 18 - At 20.0C, an aluminum ring has an inner diameter...Ch. 18 - Why is the following situation impossible? A thin...Ch. 18 - A volumetric flask made of Pyrex is calibrated at...Ch. 18 - Review. On a day that the temperature is 20.0C, a...Ch. 18 - Review. The Golden Gate Bridge in San Francisco...Ch. 18 - Your father and your younger brother are...Ch. 18 - An auditorium has dimensions 10.0 m 20.0 m 30.0...Ch. 18 - A container in the shape of a cube 10.0 cm on each...Ch. 18 - Prob. 21PCh. 18 - Prob. 22PCh. 18 - In state-of-the-art vacuum systems, pressures as...Ch. 18 - You have scored a great internship with NASA,...Ch. 18 - Review. The mass of a hot-air balloon and its...Ch. 18 - A room of volume V contains air having equivalent...Ch. 18 - Prob. 27PCh. 18 - You are applying for a position with a sea rescue...Ch. 18 - The pressure gauge on a cylinder of gas registers...Ch. 18 - A steel beam being used in the construction of a...Ch. 18 - Two metal bars are made of invar and a third bar...Ch. 18 - Why is the following situation impossible? An...Ch. 18 - A student measures the length of a brass rod with...Ch. 18 - The density of gasoline is 730 kg/m3 at 0C. Its...Ch. 18 - A liquid has a density . (a) Show that the...Ch. 18 - Prob. 36APCh. 18 - The rectangular plate shown in Figure P18.37 has...Ch. 18 - A bimetallic strip of length L is made of two...Ch. 18 - Prob. 39APCh. 18 - A vertical cylinder of cross-sectional area A is...Ch. 18 - Review. Consider an object with any one of the...Ch. 18 - Prob. 42APCh. 18 - Starting with Equation 18.11, show that the total...Ch. 18 - Review. A house roof is a perfectly flat plane...Ch. 18 - A 1.00-km steel railroad rail is fastened securely...Ch. 18 - Prob. 46CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P16.56). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find die height h in Figure P16.56. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder.arrow_forwardA manometer containing water with one end connected to a container of gas has a column height difference of 0.60 m (Fig. P15.72). If the atmospheric pressure on the right column is 1.01 105 Pa, find the absolute pressure of the gas in the container. The density of water is 1.0 103 kg/m3. FIGURE P15.72arrow_forwardA gas is in a container of volume V0 at pressure P0. It is being pumped out of the container by a piston pump. Each stroke of the piston removes a volume Vs through valve A and then pushes the air out through valve B as shown in Figure P19.74. Derive an expression that relates the pressure Pn of the remaining gas to the number of strokes n that have been applied to the container. FIGURE P19.74arrow_forward
- A manometer is shown in Figure P15.36. Rank the pressures at the five locations indicated from highest to lowest. Indicate equal pressures, if any. FIGURE P15.36arrow_forward(a) Given that air is 21% oxygen, find the minimum atmospheric pressure that gives a relatively safe partial pressure of oxygen of 0.16 atm. (b) What is the minimum pressure that gives a partial pressure of oxygen above the quickly fatal level of 0.06 atm? (c) The air pressure at the summit of Mount Everest (8848 m) is 0.334 atm. Why have a few people climbed it without oxygen, while some who have tried, even though they had trained at high elevation, had to tum back?arrow_forwardConsider the piston cylinder apparatus shown in Figure P20.81. The bottom of the cylinder contains 2.00 kg of water at just under 100.0c. The cylinder has a radius of r = 7.50 cm. The piston of mass m = 3.00 kg sits on the surface of the water. An electric heater in the cylinder base transfers energy into the water at a rate of 100 W. Assume the cylinder is much taller than shown in the figure, so we dont need to be concerned about the piston reaching the top of the cylinder. (a) Once the water begins boiling, how fast is the piston rising? Model the steam as an ideal gas. (b) After the water has completely turned to steam and the heater continues to transfer energy to the steam at the same rate, how fast is the piston rising?arrow_forward
- When a person sits erect, increasing the vertical position of their brain by 36.0 cm, the heart must continue to pump blood to the brain at the same rate. (a) What is the gain in gravitational potential energy for 100 mL of blood raised 36.0 cm? (b) What is the drop in pressure, neglecting any losses due to friction? (c) Discuss how the gain in gravitational potential energy and the decrease in pressure are related.arrow_forwardA vertical cylindrical tank contains 1.80 mol of an ideal gas under a pressure of 0.300 atm at 20.0 C. The round part of the tank has a radius of 10.0 cm, and the gas is supporting a piston that can move up and down in the cylinder without friction. There is a vacuum above the piston. (a) What is the mass of this piston? (b) How tall is the column of gas that is supporting the piston?arrow_forwardA cylinder containing ideal gas is sealed by a piston that is above the gas. The piston is a cylindrical object, with a weight of 22.0 N, which can slide up or down in the cylinder without friction. The inner radius of the cylinder, and the radius of the piston, is 8.00 cm. The top of the piston is exposed to the atmosphere, and the atmospheric pressure is 101.3 kPa. The cylinder has a height of 30.0 cm, and, when the temperature of the gas is 20°C, the bottom of the piston is 11.0 cm above the bottom of the cylinder. (A) Find the number of moles of ideal gas in the cylinder. (B) Heat is added, gradually raising the temperature of the gas to 160°C. Calculate the distance between the bottom of the cylinder and the bottom of the piston when the piston comes to its new equilibrium position.arrow_forward
- A 0.435 kg0.435 kg metal cylinder is placed inside the top of a plastic tube, the lower end of which is sealed off by an adjustable plunger. The cylinder comes to rest some distance above the plunger. The plastic tube has an inner radius of 6.56 mm6.56 mm and is frictionless. Neither the plunger nor the metal cylinder allow any air to flow around them. If the plunger is suddenly pushed upwards, increasing the pressure between the plunger and the metal cylinder by a factor of 2.792.79, what is the initial acceleration ?a of the metal cylinder? Assume the pressure outside of the tube is 1.00 atm1.00 atm and that the top of the tube is open to the air.arrow_forwardA rigid tank having a volume of 0.100 m3 contains helium gas at 175 atm. How many balloons can be inflated by opening the valve at the top of the tank? Each filled balloon is a sphere 0.300 m in diameter at an absolute pressure of 1.45 atm. (Remember that the tank cannot be completely emptied of gas.)arrow_forwardA cylinder containing ideal gas is sealed by a piston that is above the gas. The piston is a cylindrical object, with a weight of 34.0 N, which can slide up or down in the cylinder without friction. The inner radius of the cylinder, and the radius of the piston, is 6.00 cm. The top of the piston is exposed to the atmosphere, and the atmospheric pressure is 101.3 kPa. The cylinder has a height of 30.0 cm, and, when the temperature of the gas is 20°C, the bottom of the piston is 15.0 cm above the bottom of the cylinder. (a) Determine the pressure of the gas in the cylinder. kPa (b) Find the number of moles of ideal gas in the cylinder. moles (c) Heat is added, gradually raising the temperature of the gas to 185°C. Calculate the distance between the bottom of the cylinder and the bottom of the piston when the piston comes to its new equilibrium position. cmarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Thermal Expansion and Contraction of Solids, Liquids and Gases; Author: Knowledge Platform;https://www.youtube.com/watch?v=9UtfegG4DU8;License: Standard YouTube License, CC-BY