
An ideal gas with γ = 1.67 starts at point A in Fig. 18.21, where its volume and pressure are 1.00 m3 and 250 kPa, respectively. It undergoes an adiabatic expansion that triples its volume, ending at B. It’s then heated at constant volume to C, and compressed isothermally back to A. Find (a) the pressure at B, (b) the pressure at C, and (c) the net work done on the gas.
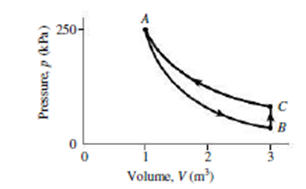
FIGURE 18.21 Problem 51
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Essential University Physics (3rd Edition)
Additional Science Textbook Solutions
The Cosmic Perspective Fundamentals (2nd Edition)
College Physics: A Strategic Approach (3rd Edition)
Conceptual Physics (12th Edition)
Conceptual Physical Science (6th Edition)
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Lecture- Tutorials for Introductory Astronomy
- A car tile contains 0.0380 m3 of air at a pressure of 2.20105 Pa (about 32 psi). How much more internal energy does this gas have than the same volume has at zero gauge pressure (which is equivalent to normal atmospheric pressure)?arrow_forwardAn ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in its internal energy and (b) its final temperature?arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forward
- Repeat the preceding calculations for an ideal diatomic gas expanding adiabatically from an initial volume of 0.500 m3 to a final volume of 1.25 m3, starting at a pressure of 1.01 105 Pa. (You must sketch the curve to find the work.) P2 = W ≈arrow_forwardA gas trapped in a piston chamber expands by 0.141 m3 at a constant pressure of 1.00 atm. If 4.28 kJ of heat are also added to the gas, by how much does the internal energy of the gas change?arrow_forwardDuring the compression stroke of a certain gasoline engine, the pressure increases from 1.00 atm to 20.0 atm. If the process is adiabatic and the air–fuel mixture behaves as a diatomic ideal gas, (a) by what factor does the volume change and (b) by what factor does the temperature change? Assuming the compression starts with 0.016 0 mol of gas at 27.0°C, find the values of (c) Q, (d) ΔEint, and (e) W that characterize the process.arrow_forward
- An ideal diatomic gas, with rotation but no oscillation, undergoes an adiabatic compression. Its initial pressure and volume are 1.20 atm and 0.200m3. Its final pressure is 2.40 atm. How much work is done by the gas?arrow_forwardSuppose a monatomic ideal gas is changed from state A to state D by one of the processes shown on the PV diagram.where P1 = 3.10 and P2 = 6.20. Find the total work done on the gas if it follows the constant-volume path AB followed by the constant-pressure path BCD.arrow_forwardSketch a PV diagram and find the work done by the gas during the following stages.(a) A gas is expanded from a volume of 1.0 L to 3.0 L at a constant pressure of 3.0atm. (b) The gas is then cooled at constant volume until the pressure falls to 2.0 atm.(c) The gas is then compressed at a constant pressure of 2.0 atm from a volume of 3.0L to 1.0 L. Note: Be careful of signs. (d) The gas is heated until its pressure increasesfrom 2.0 atm to 3.0 atm at a constant volume. (e) Find the net work done during thecomplete cycle. [203 J]arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning



