
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
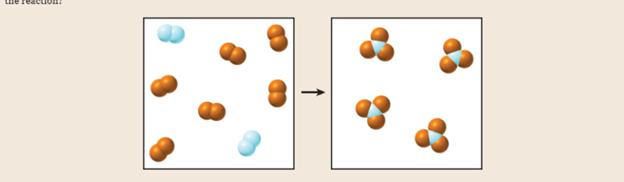
Consider the gas-phase reaction of
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Loose Leaf for Chemistry
- Predict the sign of S for each of the following and explain. a. the evaporation of alcohol b. the freezing of water c. compressing an ideal gas at constant temperature d. dissolving NaCl in waterarrow_forwardSelect the circumstances under which a reaction would most likely be spontaneous. a. delta H is positive, delta S is positive, and the temperature is low. b. delta H is positive, delta S is negative, and the temperature is low. c. delta H is positive, delta S is negative, and the temperature is high. d. delta H is negative, delta S is negative, and the temperature is high. e. delta H is negative, delta S is positive, and the temperature is high.arrow_forwardANSWER PARTS A, B, C, & D A. Which one of the following processes produces a decrease in the entropy of the system? A) boiling water to form steam B) dissolution of solid KCl in water C) mixing of two gases D) freezing water to form ice E) melting ice to form water B. ΔS is positive for the reaction __________. A) 2H2 (g) + O2 (g) → 2H2O (g) B) 2NO2 (g) → N2O4 (g) C) CO2 (g) → CO2 (s) D) BaF2 (s) → Ba2+ (aq) + 2F- (aq) E) 2Hg (l) + O2 (g) → 2HgO (s) C. For the reaction C2H6 (g) → C2H4 (g) + H2 (g) ΔH° is + 137 kJ/mol and ΔS° is +120 J/K ∙ mol. This reaction is __________. A) spontaneous at all temperatures B) spontaneous only at high temperature C) spontaneous only at low temperature D) nonspontaneous at all temperatures D. Which of these processes lead(s) to an increase in entropy? (treat all gases as ideal). Circle all that apply. A) The pressure of 1 mole of oxygen gas is allowed to double isothermally. B) Carbon dioxide is allowed to expand isothermally to 10 times its…arrow_forward
- Predict the sign of ΔS°, if possible, for the following reaction. A. N2(g) + O2(g) ----> 2NO(g) B. C2H2(g) + 2H2(g) ----> C2H6(g) Question 3 options: positive negative not predictablearrow_forwardCalculate the entropy change when graphite burns insufficient supply of oxygen as shown in the following equation, C graphite (s) + O2 (g) -> CO2 (g) Given: S° of C graphite (s) = 5.7 J/molK S° of O2 (g) = 205 J/molK S° of CO2 (g) = 213.6 J/molKarrow_forwardOut atmosphere is composed primarily of nitrogen and oxygen, which coexist at 25°C without reacting to any significant extent. However, the two gases can react to form nitrogen monoxide according to the following reaction: N2(g) + O2(g) ⇌ 2NO(g) Calculate delta G° at 1940K. (Express answer using four significant figures.) Does the reaction become spontaneous with increasing temperature?arrow_forward
- 4) For EACH process below, state whether S is positive, negative, or zero, and BRIEFLY STATE WHY. Use entropy considerations only. a) condensing ether vapor b) melting iron metal c) dissolving solid sucrose in water d) cooling N2 gas from 85 oC to 20 oCarrow_forward7.) What do you expect for the sign of Δ S in a chemical reaction in which 2 mol of gaseous reactants are converted to 3 mol of gaseous products? Group of answer choices A.) Δ S is equal to zero B.) Δ S is negative C.) Δ S is positivearrow_forwardConsider the combustion of ethane: 2C2H6 + 7O2 --> 4CO2 + 6H2O Substance ΔHf C2H6 -84.7 O2 0 CO2 -393.5 H2O -242.0 What is ΔHcombustion (ΔHreaction) ? Question 2 options: 550.8 kJ -2856.6 kJ 2856.6 kJ -550.8 kJarrow_forward
- What is the sign of Δ S for the reaction below as written? CaCO3(s) ---------> CaO(s) + CO2(g) Group of answer choices A positive B negative C zeroarrow_forwardWhat is the sign of Δ S for the reaction below as written? N2O4(g) ---------> N2(g) + 2 O2(g) Group of answer choices positive negative zeroarrow_forwardFor which set of conditions is the reaction always nonspontaneous? ΔH = + and ΔS = + ΔH = - and ΔS = - ΔH = - and ΔS = + ΔH = + and ΔS = - ΔH = 0 and ΔS = 0arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



