
Concept explainers
a)
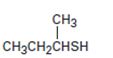
Interpretation:
The name of the compound given is to be stated.
Concept introduction:
Thioalcohols are named as derivatives of the parent
To give:
The name of the compound shown.
b)
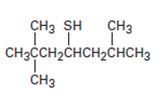
Interpretation:
The name of the compound given is to be stated.
Concept introduction:
Thioalcohols are named as derivatives of the parent alkane using the suffix –al. The longest carbon chain containing the thiol group is chosen and the parent name is derived by replacing the ending –e with –thiol. The alkane chain is numbered beginning at the end nearer to the thiol group. The substituents are numbered according to their position on the chain. The name is written listing the substituents in the alphabetical order and indicating the position to which –SH is bonded.
To give:
The name of the compound shown.
c)
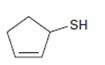
Interpretation:
The name of the compound given is to be stated.
Concept introduction:
In naming cyclic thioalcohols the parent name is derived from the cycloalkene ring by replacing –e of the cycloalkene with –thiol. The ring is numbered from the carbon with –SH in such a way that lowest number possible is given to the other functional groups present.
To give:
The name of the compound shown.
d)
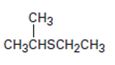
Interpretation:
The name of the compound given is to be stated.
Concept introduction:
Simple sulfides are with no other functional groups are named by identifying the two organic substituents and adding the word sulfide. If other functional groups are present, the sulfide part is considered as an alkylthio substituent.
To give:
The name of the sulfide shown.
e)
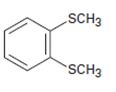
Interpretation:
The name of the compound given is to be stated.
Concept introduction:
Simple sulfides are with no other functional groups are named by identifying the two organic substituents and adding the word sulfide. If other functional groups are present, the sulfide part is considered as an alkylthio substituent.
To give:
The name of the disulfide shown.
f)
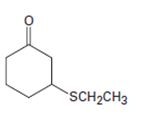
Interpretation:
The name of the compound given is to be stated.
Concept introduction:
Simple sulfides are with no other functional groups are named by identifying the two organic substituents and adding the word sulfide. If other functional groups are present, the sulfide part is considered as an alkylthio substituent.
To give:
The name of the compound shown.
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Organic Chemistry (Instructor's)
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

