
Chemistry - With Access (Looseleaf) (Custom)
3rd Edition
ISBN: 9781259896491
Author: Burdge
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18.8, Problem 1PPC
Practice Problem  CONCEPTUALIZE
CONCEPTUALIZE
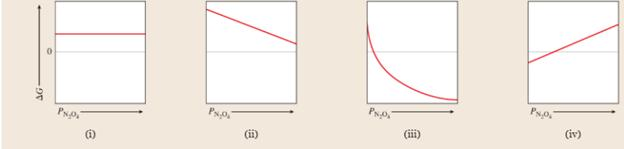
Consider the reaction in Sample Problem 18.8. Which of the following graphs best shows what happens to
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the sign of ΔS°, if possible, for the following reaction.
A. N2(g) + O2(g) ----> 2NO(g)
B. C2H2(g) + 2H2(g) ----> C2H6(g)
Question 3 options:
positive
negative
not predictable
The Kp for the reaction A (g) ⇌ 2 B (g) is 0.0730. What is Kp for the reaction 4 B (g) ⇌ 2 A (g)?
Answer numericla ______
ploA plot of ln K (equilibrium constant) for a reaction vs 1/T gave a line with slope
- 2040 kelvin and a y-intercept of 2.41. What is the standard entropy change for the
reaction?
Chapter 18 Solutions
Chemistry - With Access (Looseleaf) (Custom)
Ch. 18.1 - Practice Problem ATTEMPT
Determine the change in...Ch. 18.1 - Practice Problem BUILD To what fraction of its...Ch. 18.1 - Practice Problem CONCEPTUALIZE
Which equation is...Ch. 18.2 - Practice ProblemATTEMPT Calculate the standard...Ch. 18.2 - Practice Problem BUILD
In each of the following...Ch. 18.2 - Practice Problem CONCEPTUALIZE
For each reaction...Ch. 18.3 - Practice ProblemATTEMPT For each of the following...Ch. 18.3 - Practice Problem BUILD
Make a qualitative...Ch. 18.3 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 18.3 - 18.3.1 For which of the following physical...
Ch. 18.3 - 18.3.2 For which of the following chemical...Ch. 18.3 - 18.3.3 Identify the correct balanced equation and...Ch. 18.4 - Practice Problem ATTEMPT For each of the...Ch. 18.4 - Practice Problem BUILD (a) Calculate Δ S univ and...Ch. 18.4 - Practice Problem CONCEPTUALIZE The following table...Ch. 18.4 - Using data from Appendix 2, calculate Δ S ° (in...Ch. 18.4 - 18.4.2 Using data from Appendix 2, calculate (in...Ch. 18.4 - The diagrams show a spontaneous chemical reaction....Ch. 18.4 - 18.4.4 The diagrams show a spontaneous chemical...Ch. 18.5 - Practice Problem ATTEMPT
A reaction will be...Ch. 18.5 - Practice Problem BUILD
Given that the reaction is...Ch. 18.5 - Practice ProblemCONCEPTUALIZE Which of the...Ch. 18.5 - A reaction for which Δ H and Δ S are both negative...Ch. 18.5 - At what temperature ( in ºC ) does a reaction go...Ch. 18.5 - 18.5.3 Using data from Appendix 2, calculate G°...Ch. 18.5 - 18.5.4 Calculate for the sublimation of iodine in...Ch. 18.6 - Practice Problem ATTEMPT
Calculate the standard...Ch. 18.6 - Practice problemBUILD For each reaction, determine...Ch. 18.6 - Prob. 1PPCCh. 18.6 - 18.6.1 For the reaction:
Ch. 18.6 - Consider the reaction: X ( g ) + Y(g) ⇄ Z( g ) for...Ch. 18.6 - The Δ G° for the reaction: N 2 ( g ) + 3H 2 (g) ⇄...Ch. 18.6 - 18.6.4 The for iron(III) hydroxide . For the...Ch. 18.7 - Practice Problem ATTEMPT
The molar heats of fusion...Ch. 18.7 - Practice Problem CONCEPTUALIZE
Explain why. in...Ch. 18.8 - Practice ProblemATTEMPT Δ G ° for the reaction: H...Ch. 18.8 - Practice ProblemBUILD What is the minimum partial...Ch. 18.8 - Practice Problem CONCEPTUALIZE Consider the...Ch. 18.9 - Practice Problem ATTEMPT Using data from Appendix...Ch. 18.9 - Practice ProblemBUILD K f for the complex ion Ag (...Ch. 18.9 - Practice Problem CONCEPTUALIZE Which of the...Ch. 18.10 - Practice ProblemATTEMPT Calculate G for the...Ch. 18.10 - Practice ProblemBUILD Ksp for Co(OH)2 at...Ch. 18.10 - Prob. 1PPCCh. 18 - 18.1
Which of the following must be negative for a...Ch. 18 - Δ G for a reaction is always negative when (a) Δ G...Ch. 18 - 18.3
The diagram shown here depicts a system at...Ch. 18 - The reaction shown here has Δ G º = -1 .83 kJ/mol...Ch. 18 - 18.1 Explain what is meant by a spontaneous...Ch. 18 - Prob. 2QPCh. 18 - Prob. 3QPCh. 18 - Describe what is meant by the term entropy. What...Ch. 18 - Prob. 5QPCh. 18 - Prob. 6QPCh. 18 - Prob. 7QPCh. 18 - Prob. 8QPCh. 18 - How does the entropy of a system change for each...Ch. 18 - Prob. 10QPCh. 18 - Prob. 11QPCh. 18 - Prob. 12QPCh. 18 - Prob. 13QPCh. 18 - Using the data in Appendix 2, calculate the...Ch. 18 - 18.15 Using the data in Appendix 2, calculate the...Ch. 18 - Prob. 16QPCh. 18 - Prob. 17QPCh. 18 - Prob. 18QPCh. 18 - 18.19 State the third law of thermodynamics in...Ch. 18 - Calculate Δ S surr for each of the reactions in...Ch. 18 - Calculate Δ S surr for each of the reactions in...Ch. 18 - Using data from Appendix 2, calculate Δ S rxn º...Ch. 18 - 18.23 Using data from Appendix 2, calculate for...Ch. 18 - Prob. 24QPCh. 18 - Why is it more convenient to predict the direction...Ch. 18 - What is the significance of the sign of Δ G sys ?Ch. 18 - From the following combinations of Δ H and Δ S ,...Ch. 18 - Prob. 28QPCh. 18 - Prob. 29QPCh. 18 - From the values of Δ H and Δ S , predict which of...Ch. 18 - Find the temperatures at which reactions with the...Ch. 18 - The molar heats of fusion and vaporization of...Ch. 18 - 18.33 The molar heats of fusion and vaporization...Ch. 18 - Prob. 34QPCh. 18 - Prob. 35QPCh. 18 - Prob. 36QPCh. 18 - Prob. 37QPCh. 18 - Prob. 38QPCh. 18 - Explain why Equation 18.14 is of great importance...Ch. 18 - Prob. 40QPCh. 18 - Prob. 41QPCh. 18 - Prob. 42QPCh. 18 - 18.43 Consider the following reaction at...Ch. 18 - Prob. 44QPCh. 18 - 18.45
(a)
Calculate and for the following...Ch. 18 - Prob. 46QPCh. 18 - Consider the decomposition of calcium carbonate:...Ch. 18 - Prob. 48QPCh. 18 - 18.49 At for the process:
is 8.6 kJ/mol....Ch. 18 - Prob. 50QPCh. 18 - What is a coupled reaction? What is its importance...Ch. 18 - What is the role of ATP in biological reactions?Ch. 18 - Prob. 53QPCh. 18 - 18.54 In the metabolism of glucose, the first step...Ch. 18 - Predict the signs of Δ H , Δ S , and Δ G of the...Ch. 18 - Prob. 56APCh. 18 - Prob. 57APCh. 18 - Prob. 58APCh. 18 - Prob. 59APCh. 18 - Prob. 60APCh. 18 - Ammonium nitrate ( NH 4 NO 3 ) dissolves...Ch. 18 - 18.62 Calculate the equilibrium pressure of due...Ch. 18 - Prob. 63APCh. 18 - Referring to Problem 18.63, explain why the ratio...Ch. 18 - 18.65 Which of the following are not state...Ch. 18 - 18.66 For reactions carried out under...Ch. 18 - Prob. 67APCh. 18 - Prob. 68APCh. 18 - A student looked up the Δ G f o , Δ H f o , and Δ...Ch. 18 - Consider the following Brønsted acid-base reaction...Ch. 18 - 18.71 At o K, the entropy of carbon monoxide...Ch. 18 - Prob. 72APCh. 18 - Consider the thermal decomposition of CaCO 3 :...Ch. 18 - Prob. 74QPCh. 18 - Prob. 75QPCh. 18 - Prob. 76QPCh. 18 - Prob. 77APCh. 18 - Prob. 78APCh. 18 - Prob. 79APCh. 18 - Prob. 80APCh. 18 - Prob. 81APCh. 18 - Prob. 82APCh. 18 - 18.83 Comment on the statement: “Just talking...Ch. 18 - Prob. 84APCh. 18 - Consider the reaction: N 2 ( g ) + O 2 ( g ) ⇄ 2...Ch. 18 - Prob. 86APCh. 18 - Consider the decomposition of magnesium carbonate:...Ch. 18 - Prob. 88APCh. 18 - Prob. 89APCh. 18 - 18.90 The rate constant for the elementary...Ch. 18 - A 74.6-g ice cube floats in the Arctic Sea. The...Ch. 18 - 18.92 Which of the following is not accompanied by...Ch. 18 - Prob. 93APCh. 18 - Give a detailed example of each of the following,...Ch. 18 - Prob. 95QPCh. 18 - 18.96 The standard enthalpy of formation and the...Ch. 18 - Prob. 97QPCh. 18 - Prob. 98QPCh. 18 - The following reaction was described as the cause...Ch. 18 - Comment on the feasibility of extracting copper...Ch. 18 - 18.101 One of the steps in the extraction of iron...Ch. 18 - Prob. 102APCh. 18 - Prob. 103APCh. 18 - Prob. 104APCh. 18 - 18.105 The enthalpy change in the denaturation of...Ch. 18 - Prob. 106APCh. 18 - Prob. 107APCh. 18 - Prob. 108APCh. 18 - Prob. 109APCh. 18 - Prob. 110APCh. 18 - 18.111 Carbon monoxide and nitric oxide are...Ch. 18 - Prob. 112APCh. 18 - Prob. 113APCh. 18 - 18.114 Many hydrocarbons exist as structural...Ch. 18 - Physical and Biological Sciences
In chemistry, the...Ch. 18 - Physical and Biological Sciences
In chemistry, the...Ch. 18 - Prob. 3SEPPCh. 18 - Physical and Biological Sciences
In chemistry, the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the reactionHCl(g)+ NH3(g)NH4Cl(s)Using the standard thermodynamic data in the tables linked above, calculate the equilibrium constant for this reaction at 298.15K.ANSWER:arrow_forwardConsider the reactionI2(g) + Cl2(g)2ICl(g)Using the standard thermodynamic data in the tables linked above, calculate the equilibrium constant for this reaction at 298.15K.ANSWER:arrow_forwarddetermine the Kp value for: SO2(g) + 1/2 O2(g) ⇌ SO3(g) Answer: Kp = 1.9 atm-1/2 *I know how to get the answer, however I don't know how the unit is atm-1/2 , please explain step-by-step.arrow_forward
- Consider the reaction2SO2(g) + O2(g)->2SO3(g)Using the standard thermodynamic data in the tables linked above, calculate deltaGrxn for this reaction at 298.15K if the pressure of each gas is 39.97 mm Hg.ANSWER: kJ/molarrow_forwardSolve all part please. Consider the equilibrium reaction N2O4 (g) ⇌ 2 NO2 (g)a. Using appendix C, find H and S for this reaction (you will need this for b,c). delta s= 176.6J delta H=5802kJb. Knowing that K is temperature dependent and that G = -RTlnK, find the equilibrium constant (K) and T whenthe above reaction is at equilibrium with PNO2 = PN2O4 = 1.0 atm.c. Knowing that K is temperature dependent and that G = -RTlnK, find the equilibrium constant (K) and T whenthe above reaction is at equilibrium with PT = 10.0 atm and PNO2 = 2 PN2O4.arrow_forwardIn the gas-phase reaction A + B = C + 2 D, it was found that, when 2.00 mol A, 1.00 mol B and 3.00 mol D were mixed and allowed to come to equilibrium at 25oC, the resulting mixture contained 0.79 mol C at a total pressure of 1.00 bar. Calculate Kp. Answer: [.960]arrow_forward
- 2.Consider the reaction H2(g) + C2H4(g)C2H6(g) Using the standard thermodynamic data in the tables , calculate Grxn for this reaction at 298.15K if the pressure of each gas is 39.55 mm Hg. ANSWER:____ kJ/molarrow_forwardConsider the following reaction at 298K.Cu2+ (aq) + Cr2+ (aq) Cu+ (aq) + Cr3+ (aq)Which of the following statements are correct?Choose all that apply. Eocell > 0 The reaction is reactant-favored. n = 4 mol electrons delta Go > 0 K > 1 Submit Answerarrow_forwardFor which of the following is the change in entropy positive? Check all that apply. Explain. A. C 6 H 12 (l) C 6 H 12 (g)B. Dilution of 1.0 mL of 1.0 M NaOH with 90 mL of waterC. . 2 O 2 (g) + 2 SO (g) 2 SO 3 (g)D. grinding a large crystal of NaCl to powderE. Cooling water from room temperature to 15°C.arrow_forward
- What is K if ΔG° = -20.0 kJ for a reaction at 25°? (SHOW CALCULATION)arrow_forwardSOLVE THE FOLLOWING PROBLEMS. Write down your answers CLEARLY. Thank you! Lesson Topic: THERMODYNAMICS (Spontaneous reactions and Gibbs free energy) 1. Use the given standard entropy values to calculate the standard entropy change to convert hydrogen chloride to chlorine in the given reaction: 4HCl ₍g₎ + O2₍g₎ → 2Cl₂₍g₎ + 2H₂O₍g₎ S°(J/K•mol): HCl ₍g₎ = 186.8 O2₍g₎ = 205 Cl2₍g₎ = 223 H2O₍g₎ = 188.7 2. Predict whether the following reaction leads to an increase or a decrease in entropy. Explain Why. Ag+₍aq₎ + Cl-₍aq₎ → AgCl₍s₎ 3. Calculate ∆G° for the combustion of ethane 2C₂H₆₍g₎ + 7O₂₍g₎ → 4CO₂₍g₎ + 6H₂O₍ₗ₎ With the following ∆G°f: C₂H₆(g = -32.86 kJ/mol CO₂(g) = -394.4 kJ/mol H₂O(l) = -237.2 kJ/mol O₂(g) = 0 4. From the following ∆H and ∆S values, predict whether the reaction would be spontaneous. If not, at what temperature…arrow_forwardCalculate K at 298 K for the following reaction: NO(g) + 1/2 O2 (g) <-> NO2 (g) Delta G (kJ/mol) for the following: NO(g) 86.60, NO2(g) 51, O2(g) 0 (answer in scientific notation)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY