
Concept explainers
Draw the structure corresponding to each name.
(a)
(b)
(c)
(d)
(e) potassium acetate
(f) sodium
(g)
(h)
(a)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to
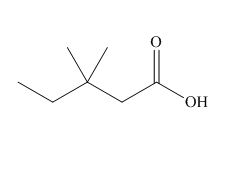
Explanation of Solution
The name of the compound is

Figure 1
The structure of
(b)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to
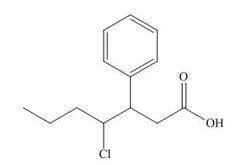
Explanation of Solution
The name of the compound is
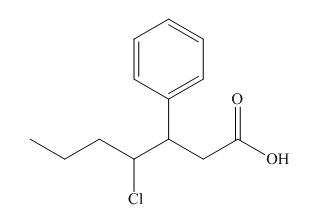
Figure 2
The structure of
(c)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to
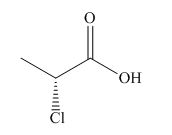
Explanation of Solution
The name of the compound is
The stereochemistry of the compound is determined by prioritizing the groups attached to its stereogenic center. The groups are prioritized on the basis of atomic number of their atoms. The configuration of the compound is
Therefore, the structure corresponding to the given name is,

Figure 3
The structure of
(d)
Interpretation: The structure corresponding to given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to
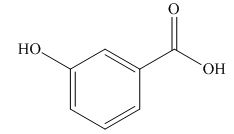
Explanation of Solution
The name of the compound is

Figure 4
The structure of
(e)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to potassium acetate is,
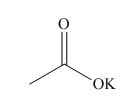
Explanation of Solution
The name of the compound is potassium acetate. The name suggests that potassium is present in the place of hydrogen in acetic acid. Therefore, the structure corresponding to the given name is,

Figure 5
The structure of potassium acetate is drawn in Figure 5.
(f)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to sodium
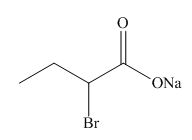
Explanation of Solution
The name of the compound is sodium

Figure 6
The structure of sodium
(g)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to
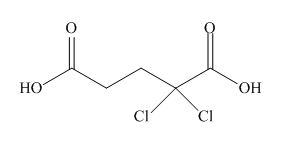
Explanation of Solution
The name of the compound is

Figure 7
The structure of
(h)
Interpretation: The structure corresponding to the given name is to be stated.
Concept introduction: International Union of Pure and Applied Chemistry gave guidelines to write the chemical name of molecules. There are three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of the parent name and the functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is the addition of substituents at appropriate carbon atoms.
Answer to Problem 19.30P
The structure corresponding to
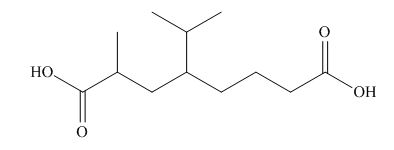
Explanation of Solution
The name of the compound is

Figure 8
The structure of
Want to see more full solutions like this?
Chapter 19 Solutions
ORGANIC CHEMISTRY(LL)W/ACCESS>CUSTOM<
- The Ka1 of ascorbic acid is 7.94 x 10-5. Would you expect ascorbic acid dissolved in blood plasma (pH 7.35–7.45) to exist primarily as ascorbic acid or as ascorbate anion? Explain.arrow_forwardWhich of the following is the correct order of basicity of amines? A. B > C > AB. A > C > BC. B > A > CD. C > A > BE. A > B > Carrow_forwardRank the nucleophilicity of the 3 bases: Na2CO3, K2cO3, NaOHarrow_forward
- Although it was initially sold as a rat poison, warfarin is an effective anticoagulant used to prevent blood clots. Label the most acidic proton in warfarin, and explain why its pKa is comparable to the pKa of a carboxylic acid.arrow_forwardRank each set of compounds in order of increasing basicity.aniline, p-methylaniline, p-nitroanilinearrow_forwardThe hydrolysis of the ester shown here is catalyzed by morpholine. Explain how morpholine catalyzes the reaction. (Hint: The pKa of the conjugateacid of morpholine is 9.3, so morpholine is too weak a base to function as a base catalyst.)arrow_forward
- Arrange the members of each group in order of decreasing basicity: (a) Ammonia, aniline, methylamine (b) Acetanilide, aniline, N-methylaniline (c) 2,4-Dichloroaniline, 2,4-dimethylaniline, 2,4-dinitroaniline (d) 3,4-Dichloroaniline, 4-chloro-2-nitroaniline, 4-chloro-3-nitroaniline (e) Dimethylamine, diphenylamine, N-methylanilinearrow_forwardCompound X is optically inactive and has the formula C 16H 16Br 2. On treatment with strong base, X gives hydrocarbon Y, C 16H 14. Compound Y absorbs 2 equivalents of hydrogen when reduced over a palladium catalyst and reacts with ozone to give two fragments. One fragment Z, is an aldehyde with formula C 7H 6O. The other fragment is glyoxal, (CHO)2. Which of the following answers is correct? Select all that are correct.arrow_forward. Explain how you would rank the amines given below according to their basicity, together with the reasons.CH3NH2 (CH3)2NH (CH3)3Narrow_forward
- (1) Which is the most basic amine and which is a secondary amine? (2) Which can undergo hydrolysis? (3) Which gives a diazonium salt upon reaction with HNO2, HCl at 0oC?arrow_forwardThe formula for the danger sign pheromone for a species of ant is C7H7O. When treated with I2 NaOH, this pheromone produces iodoform and n-hexanoic acid. How is this pheromone structure?arrow_forwardOf the following compounds, rank each series by nucleophilicity using 1 for the most nucleophilic compound and 5 for the most basic compound.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

