
Concept explainers
Rank the compounds in each group in order of increasing basicity.
a.
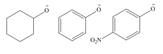
b.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry (Looseleaf)
- Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC≡CH, (CF3)2CHOH, CH3OH (b) Phenol, p-methylphenol, p-[trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxybenzonic acidarrow_forward1. Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2O2. Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds, and explain why the acidity increases so dramatically with substitution by nitro groups. CH4 CH3NO2 CH2(NO2)2 CH(NO2)3arrow_forwardArrange the given compounds based on their relative Brønsted acidities. H-F H-Br H-SH H-CH3 H-NH2arrow_forward
- Which of the following is the strongest acid? Group of answer choices CH3OH CH3OH2+ CH3NH2 CH3NH3+arrow_forwardDraw the structure of a constitutional isomer of compound B that fits each description. an isomer that is at least 105 times more acidic than B an isomer that is at least 105 times less acidic than B an isomer that is comparable in acidity to Barrow_forwardAnswer the following questions about esmolol, a drug used to treat high blood pressure sold under the trade name Brevibloc.a.Label the most acidic hydrogen atom in esmolol. b.What products are formed when esmolol is treated with NaH? c.What products are formed when esmolol is treated with HCl? d. Label all sp2 hybridized C atoms. e.Label the only trigonal pyramidal atom. f.Label all C's that bear a δ+ charge.arrow_forward
- Answer the following questions about esmolol, a drug used to treat high blood pressure sold under the trade name Brevibloc.a.Label the most acidic hydrogen atom in esmolol. b.What products are formed when esmolol is treated with NaH? c. What products are formed when esmolol is treated with HCl? d.Label all sp2 hybridized C atoms. e. Label the only trigonal pyramidal atom. f. Label all C's that bear a δ+ charge.arrow_forwardA) for each compound show its conjugate base. lone pairs have been left out. B) rank the conjugate base in the order you would predict, from most to least stable. C) rank the original compounds in order, from strongest to weakest acid.arrow_forwardGiven the following acid-base equilibria:HCOOH + H2O ---> H3O+ + HCOO-CH3NH3+ + H2O---> H3O+ + CH3NH2HSO4- + H2O---> H3O+ + SO42-Circle strongest acid and cross out the weakest.HCOOH HSO4- CH3NH3+Circle strongest base and cross out the weakest.HCOO- SO2- CH3NH2arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

