
(II) Consider the following two-step process. Heat is allowed to flow out of an ideal gas at constant volume so that its pressure drops from 2.2 atm to 1.4 atm. Then the gas expands at constant pressure, from a volume of 5.9 L to 9.3 L, where the temperature reaches its original value. See Fig. 19-30. Calculate (a) the total work done by the gas in the process, (b) the change in internal energy of the gas in the process, and (c) total heat flow into out of the gas.
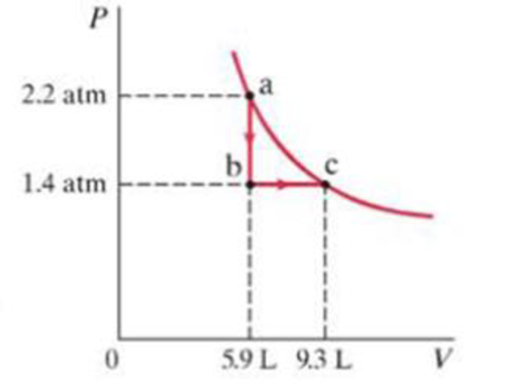
FIGURE 19-30
Problem 31.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Modified Mastering Physics With Pearson Etext -- Standalone Access Card -- For Physics For Scientists & Engineers With Modern Physics (5th Edition)
Additional Science Textbook Solutions
Conceptual Physics (12th Edition)
Conceptual Integrated Science
Physics (5th Edition)
Modern Physics
University Physics with Modern Physics (14th Edition)
The Cosmic Perspective (8th Edition)
- Compare the charge in internal energy of an ideal gas for a quasi-static adiabatic expansion with that for a quasi-static isothermal expansion. What happens to the temperature of an ideal gas in an adiabatic expansion?arrow_forwardTwo moles of nitrogen gas, with =7/5 for ideal diatomic gases, occupies a volume of 102 m3 in an insulated cylinder at temperature 300 K. The gas is adiabatically and reversibly compressed to a volume of 5 L. The piston of the cylinder is locked in its place, and the insulation around the cylinder is removed. The heat-conducting cylinder is then placed in a 300-K bath. Heat from the compressed gas leaves the gas, and the temperature of the gas becomes 300 K again. The gas is then slowly expanded at the fixed temperature 300 K until the volume of the gas becomes 102 m3, thus making a complete cycle for the gas. For the entire cycle, calculate (a) the work done by the gas, (b) the heat into or out of the gas, (c) the change in the internal energy of the gas, and (d) the change in entropy of the gas.arrow_forwardA cylinder containing three moles of a monatomic ideal gas is heated at a constant pressure of 2 atm. The temperature of the gas changes from 300 K to 350 K as a result of the expansion. Find work done (a) on the gas; and (b) by the gas.arrow_forward
- A car tile contains 0.0380 m3 of air at a pressure of 2.20105 Pa (about 32 psi). How much more internal energy does this gas have than the same volume has at zero gauge pressure (which is equivalent to normal atmospheric pressure)?arrow_forwardAn amount of n moles of a monatomic ideal gas in a conducting container with a movable piston is placed in a large thermal heat bath at temperature T1 and the gas is allowed to come to equilibrium. After the equilibrium is leached, the pressure on the piston is lowered so that the gas expands at constant temperature. The process is continued quasi-statically until the final pressure is 4/3 of the initial pressure p1 . (a) Find the change in the internal energy of the gas. (b) Find the work done by the gas. (c) Find the heat exchanged by the gas, and indicate, whether the gas takes in or gives up heat.arrow_forwardThe energy output of a heat pump is greater than the energy used to operate the pump. Why doesn't this statement violate the first law of thermodynamics?arrow_forward
- It is found that, when a dilute gas expands quasi-statically from 0.50 to 4.0 L, it does 250 J of work. Assuming that the gas temperature remains constant at 300 K, how many moles of gas are present?arrow_forward(II) 8.5 moles of an ideal monatomic gas expand adiabatically, performing 8300 J of work in the process. What is the change in temperature of the gas during this expansion?arrow_forwardii) Predict the heat taken by a gas, if the gas expands from 1100 cm³ to 2600 cm³ at constant pressure of 30000 Pa and the internal energy increases by 23 J.arrow_forward
- ii) Predict the heat taken by a gas, if the gas expands from 200 cm to 450 cm at constant pressure of 120000 Pa and the internal energy increases by 30 J.arrow_forward. (II) A gas is enclosed in a cylinder fitted with a lightfrictionless piston and maintained at atmospheric pressure.When 254 kcal of heat is added to the gas, the volumeis observed to increase slowly from 12.0 m3 to 16.2 m3Calculate (a) the work done by the gas and (b) the changein internal energy of the gas.arrow_forward9. (II) 8.5 moles of an ideal monatomic gas expand adiabat- ically, performing 8300 J of work in the process. What is the change in temperature of the gas during this expansion? 10. (II) Consider the following two-step process. Heat is allowed to flow out of an ideal gas at constant volume so that its pres- sure drops from 2.2 atm to 1.4 atm. Then the gas expands at constant pressure, from a volume of 5.9 L to 9.3 L, where the temperature reaches its original value. See Fig. 15-22. Calculate (a) the total work done by the gas in the process, (b) the change in internal P of the gas in energy of the the process, and (c) the total heat flow into or out of the gas. FIGURE 15-22 Problem 10. 2.2 atm 1.4 atm 0 b a 5.9 L 9.3 L Varrow_forward

 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning




