
a)
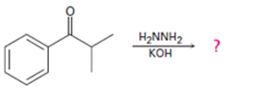
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism, beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
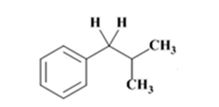
The electron-pushing mechanism for the formation of the alkane, beginning from the hydrazone intermediate, is given below.
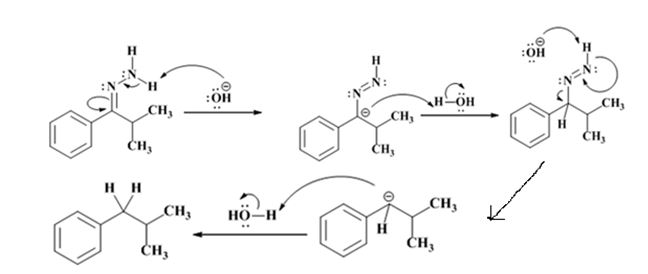
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of isopropyl phenyl ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane, ethyl benzene, as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism, beginning from the hydrazone intermediate is given below.

b)
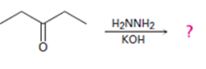
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction aldehydes and ketones react with hydrazine in the presence of a base to yield alkanes. First a hydrazone is formed which is then converted into an alkane.
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism the formation of the alkane beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
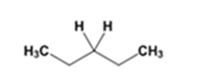
The electron-pushing mechanism the formation of the alkane, beginning from the hydrazone intermediate, is given below.
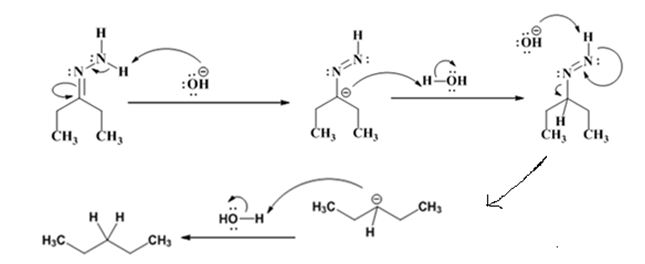
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of diethyl ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane, n-pentane, as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism, beginning from the hydrazone intermediate is given below.

c)
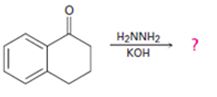
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction aldehydes and ketones react with hydrazine in the presence of a base to yield alkanes. First a hydrazone is formed which is then converted into an alkane.
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism the formation of the alkane beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
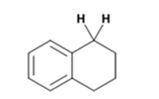
The electron-pushing mechanism the formation of the alkane, beginning from the hydrazone intermediate, is given below.
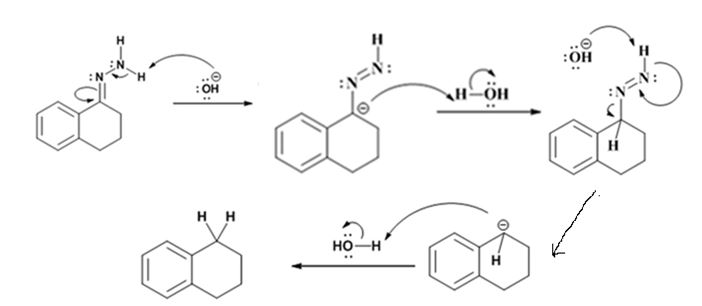
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of the ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism, beginning from the hydrazone intermediate is given below.

d)
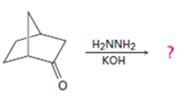
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction aldehydes and ketones react with hydrazine in the presence of a base to yield alkanes. First a hydrazone is formed which is then converted into an alkane.
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism, beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
The electron-pushing mechanism for the formation of the alkane beginning from the hydrazone intermediate, is given below.
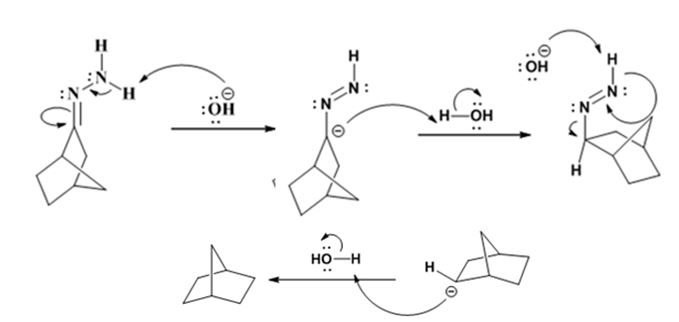
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism for the formation of the alkane beginning from the hydrazone intermediate, is given below.

Want to see more full solutions like this?
Chapter 19 Solutions
ORGANIC CHEMISTRY(LL)+OWLV2 4 TERM>BI<
- Following is a retrosynthetic scheme for the synthesis of the tricyclic diene on the left. Show how to accomplish this synthesis from 2-bromopropane, cyclopentadiene, and 2-cyclohexenone.arrow_forwarda-Tetralone undergoes Birch reduction to give an excellent yield of a single product. Predict the structure of the product, and propose a mechanism for its formation.arrow_forwardSuggest starting materials for the formation of the following Claisen condensation products:arrow_forward
- Predict the products of the reaction of the below reagents with either acetophenone or phenylacetaldehyde. NaBH4, H3O+ HCN, KCN H2NNH2 CH3MgBr, then H3O+ NH2OH, catalytic acidarrow_forwardGive the expected products from the following reactionsarrow_forwardLike other hydroborations, the reaction of alkynes with catecholborane is a syn addition and its regioselectivity is opposite to Markovnikov’s rule.Use this fact to outline a synthesis of the compound shown from 1-hexyne and (Z)-C6H5CH=CHBr.arrow_forward
- Predict the major products of treating the following compounds with hot, concentratedpotassium permanganate, followed by acidification with dilute HCl. (tetralin)arrow_forwardPredict the products of NaBH4 reduction of the following compounds?arrow_forwardPropose a mechanism for the acid-catalyzed hydration of propene. Remember that a mechanism must include curved arrows to show movement of electrons, as well as all intermediates.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


