
a)
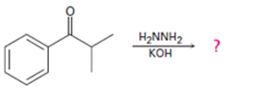
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism, beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
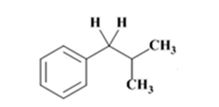
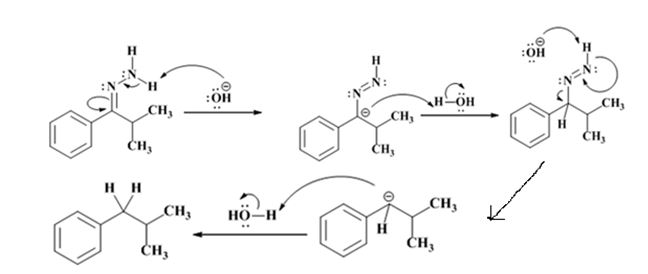
The electron-pushing mechanism for the formation of the alkane, beginning from the hydrazone intermediate, is given below.
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of isopropyl phenyl ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane, ethyl benzene, as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism, beginning from the hydrazone intermediate is given below.

b)
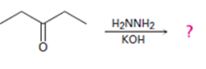
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction aldehydes and ketones react with hydrazine in the presence of a base to yield alkanes. First a hydrazone is formed which is then converted into an alkane.
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism the formation of the alkane beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
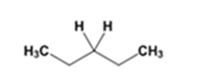
The electron-pushing mechanism the formation of the alkane, beginning from the hydrazone intermediate, is given below.
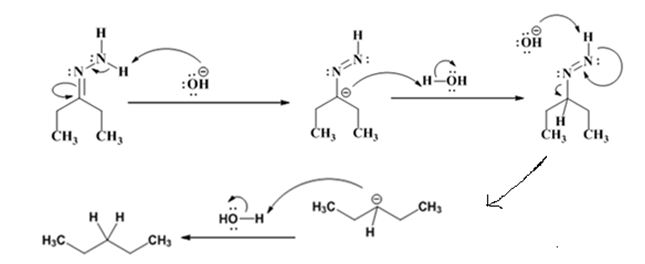
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of diethyl ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane, n-pentane, as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism, beginning from the hydrazone intermediate is given below.

c)
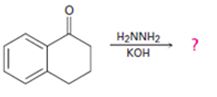
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction aldehydes and ketones react with hydrazine in the presence of a base to yield alkanes. First a hydrazone is formed which is then converted into an alkane.
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism the formation of the alkane beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
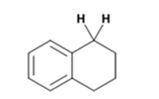
The electron-pushing mechanism the formation of the alkane, beginning from the hydrazone intermediate, is given below.
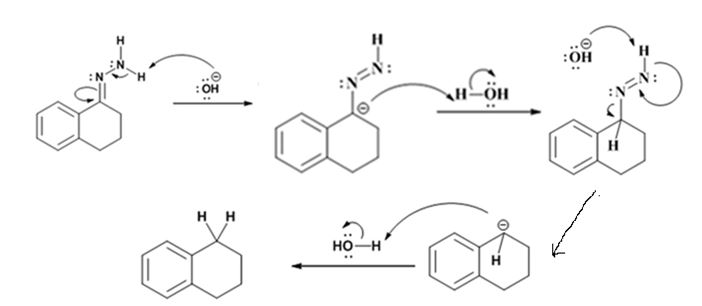
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of the ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism, beginning from the hydrazone intermediate is given below.

d)
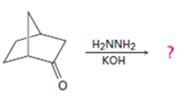
Interpretation:
The product of the Wolff-Kishner reduction reaction shown is to be given. The electron-pushing mechanism, beginning from the hydrazone intermediate is to be provided.
Concept introduction:
In Wolff-Kishner reduction aldehydes and ketones react with hydrazine in the presence of a base to yield alkanes. First a hydrazone is formed which is then converted into an alkane.
To give:
The product of the Wolff-Kishner reduction reaction shown.
To provide:
The electron-pushing mechanism, beginning from the hydrazone intermediate.
Answer to Problem 38MP
The product of the Wolff-Kishner reduction reaction shown is
The electron-pushing mechanism for the formation of the alkane beginning from the hydrazone intermediate, is given below.
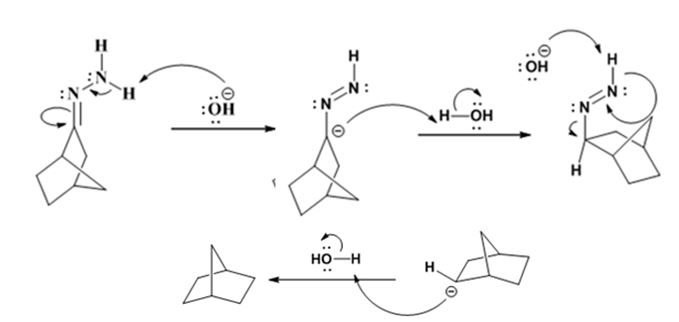
Explanation of Solution
The hydroxide ion from KOH abstracts a weakly acidic H from –NH2 of the hydrazone of ketone to yield a carbanion which picks up a proton to yield a neutral intermediate. Deprotonation of the remaining hydrogen on N by the hydroxide ion occurs with the eliminartion of nitrogen to yield another carbanion which is protonated to give the alkane as the product.
The product of the Wolff-Kishner reduction reaction shown is

The electron-pushing mechanism for the formation of the alkane beginning from the hydrazone intermediate, is given below.

Want to see more full solutions like this?
Chapter 19 Solutions
Study Guide with Student Solutions Manual for McMurry's Organic Chemistry, 9th
- Following is a retrosynthetic scheme for the synthesis of the tricyclic diene on the left. Show how to accomplish this synthesis from 2-bromopropane, cyclopentadiene, and 2-cyclohexenone.arrow_forwarda-Tetralone undergoes Birch reduction to give an excellent yield of a single product. Predict the structure of the product, and propose a mechanism for its formation.arrow_forwardSuggest starting materials for the formation of the following Claisen condensation products:arrow_forward
- Predict the products of the reaction of the below reagents with either acetophenone or phenylacetaldehyde. NaBH4, H3O+ HCN, KCN H2NNH2 CH3MgBr, then H3O+ NH2OH, catalytic acidarrow_forwardGive the expected products from the following reactionsarrow_forwardLike other hydroborations, the reaction of alkynes with catecholborane is a syn addition and its regioselectivity is opposite to Markovnikov’s rule.Use this fact to outline a synthesis of the compound shown from 1-hexyne and (Z)-C6H5CH=CHBr.arrow_forward
- Predict the major products of treating the following compounds with hot, concentratedpotassium permanganate, followed by acidification with dilute HCl. (tetralin)arrow_forwardPredict the products of NaBH4 reduction of the following compounds?arrow_forwardPropose a mechanism for the acid-catalyzed hydration of propene. Remember that a mechanism must include curved arrows to show movement of electrons, as well as all intermediates.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


