
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
12th Edition
ISBN: 9781119304241
Author: Solomons
Publisher: WILEY C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 14PP
Practice Problem 2.14
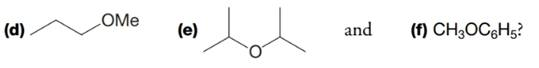
One way of naming ethers is to name the two alkyl groups attached to the oxygen atom in alphabetical order and add the word ether. If the two alkyl groups are the same, we use the prefix di-, for example, as in dimethyl ether. Write bond-line structural formulas for (a) diethyl ether, (b) ethyl propyl ether, and (c) ethyl isopropyl ether. What name would you give to
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(c) There is one (1) other positional isomer of X missing for reaction II. Draw the structural formula of this isomer.
An alkane, P, has the molecular formula, C,H.. An alkene, Q, has the
molecular formula, C H,.
(a) Name P and Q ánd write their full structural formulae.
(b) State two differences between P and Q in terms of their structures.
x'
The skeletal line formula for a branched alkene is shown below. (i) What is the molecular formula of this compound? (ii) How many carbon atoms are in the longest chain, ignoring the double bond? (iii) What is the longest chain incorporating both carbons of the double bond? (iv) How many substituents are on this chain? (v) Give the IUPAC name for this compound. [6]
Chapter 2 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Ch. 2 - Practice Problem 2.1
Propose structures for two...Ch. 2 - Prob. 2PPCh. 2 - Prob. 3PPCh. 2 - Prob. 4PPCh. 2 - Prob. 5PPCh. 2 - Practice Problem 2.6
Using a three-dimensional...Ch. 2 - Practice Problem 2.7
Trichloromethane (, also...Ch. 2 - Prob. 8PPCh. 2 - Prob. 9PPCh. 2 - Practice Problem 2.10
Write bond-line structural...
Ch. 2 - Practice Problem 2.11 Although we shall discuss...Ch. 2 - Practice Problem 2.12 Write bond-line structural...Ch. 2 - Prob. 13PPCh. 2 - Practice Problem 2.14
One way of naming ethers is...Ch. 2 - Practice Problem 2.15 Eugenol is the main...Ch. 2 - Practice Problem 2.16
One way of naming amines is...Ch. 2 - Practice Problem 2.17 Which amines in Practice...Ch. 2 - Prob. 18PPCh. 2 - Prob. 19PPCh. 2 - Practice Problem 2.20
Write bond-line formulas for...Ch. 2 - Practice Problem 2.21
Write bond-line formulas for...Ch. 2 - Practice Problem 2.22
Write bond-line formulas for...Ch. 2 - Prob. 23PPCh. 2 - Practice Problem 2.24 Write another resonance...Ch. 2 - Prob. 25PPCh. 2 - Practice Problem 2.26
Which compound would you...Ch. 2 - Practice Problem 2.27 Arrange the following...Ch. 2 - Prob. 28PPCh. 2 - Prob. 29PCh. 2 - Identify all of the functional groups in each of...Ch. 2 - 2.31 There are four alkyl bromides with the...Ch. 2 - Prob. 32PCh. 2 - Classify the following alcohols as primary,...Ch. 2 - 2.34 Classify the following amines as primary,...Ch. 2 - Prob. 35PCh. 2 - Identify all of the functional groups in Crixivan,...Ch. 2 - 2.37 Identify all of the functional groups in...Ch. 2 - 2.38 (a) Indicate the hydrophobic and hydrophilic...Ch. 2 - Hydrogen fluoride has a dipole moment of 1.83 D;...Ch. 2 - 2.40 Why does one expect the cis isomer of an...Ch. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - 2.44 Consider each of the following molecules in...Ch. 2 - True or false: For a molecule to be polar, the...Ch. 2 - 2.46 Which compound in each of the following...Ch. 2 - Prob. 47PCh. 2 - The IR spectrum of propanoic acid (Fig. 2.16)...Ch. 2 - Prob. 49PCh. 2 - Write structural formulas for four compounds with...Ch. 2 - There are four amides with the formula C3H7NO. (a)...Ch. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - 2.56 Compound C is asymmetric, has molecular...Ch. 2 - 2.57 Examine the diagram showing an -helical...Ch. 2 - Prob. 1LGPCh. 2 - Prob. 2LGPCh. 2 - Prob. 3LGPCh. 2 - Consider the molecular formula C4H8O2. Predict...Ch. 2 - Consider the molecular formula C4H8O2. If any of...Ch. 2 - Prob. 6LGPCh. 2 - Consider the molecular formula.
7. Pick five...Ch. 2 - Prob. 8LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
4.7'6. Methanol is produced by reacting carbon monoxide and hydrogen. A fresh feed stream containing CO and H2 ...
Elementary Principles of Chemical Processes, Binder Ready Version
Determine whether adding an acid will increase the solubility of the following salts: (a)AgBr,(b)CaCO3,(c)Ag2Cr...
Chemistry: The Molecular Nature of Matter
Determine the Coulombic and exchange energies for the following states, and determinewhich state is favored (ha...
Inorganic Chemistry
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Q8. Perform the calculation to the correct number of significant figures.
a) 0.121
b) 0.12
c) 0.12131
d) 0.121...
Chemistry: A Molecular Approach
6.1 State the number of electrons that be must be lost by atoms of each of the following to achieve a stable el...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- This question is about the chemistry of alkenes, which are unsaturated hydrocarbons. (a) State what is meant by the term unsaturated as applied to a hydrocarbon. (1) (b) An organic compound, X, is an unsaturated hydrocarbon with molecular formula CH₂. (i) Draw the displayed formulae and give the names of two molecules with molecular formula C₂H, which are E/Z isomers. (3) Isomer 1 Isomer 2 Name: Name:arrow_forwardProblem 2.12 Which cycloalkanes show cis,trans isomerism? For each that does, draw both isomers. (a) (b) (c)arrow_forward(A) A researcher analyzed an unknown solid, extracted from the bark of spruce trees, to determine its percentage composition. An 11.32 mg sample was burned in a combustion apparatus. The carbon dioxide (24.87 mg) and water (5.82 mg) were collected and weighed. From the results of this analysis, Determine: (1) both empirical and molecular formulas if molar mass is 420 g/mol. (ii) How many aromatic rings can this compound contain? (iii)Give the possible chemical name and structure of this compound and hence reasons for your selection.arrow_forward
- 1. (A) DRAW are all the possible isomers for dibromobutane and the cylic isomers for C3H4Br2 (B) WRITE THE STRUCTURAL OR CONDENSED FORMULAR FOR C3H7Br C3H6Br2 C3H5Br3 C5H11Brarrow_forwardThe functional class IUPAC name of the compound show below is . (You may consider to type the name in a text editor, check for its correctness and paste it in the blank.) Brarrow_forwardGive the molecular, structural and displayed formulae of the following molecules: (a) 2,2-dimethylpropane (b) 3-bromo-2, 4, 4-trimethyloctanearrow_forward
- Draw structures that t each description and name the functional group in each molecule: (a) two constitutional isomers with molecular formula C5H10O that contain different functional groups; (b) two constitutional isomers with molecular formula C6H10O that contain the same functional group.arrow_forwardDraw the structural formulas for the following compounds. Include all the bonds to hydrogen atoms. Be sure to answer both parts. (a) 1,2,4-trimethylbenzene: (b) chlorobenzene:arrow_forwardIdentify which of the following statement(s) is/are true. (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one -OH group, a secondary alcohol has two -OH groups, and a tertiary alcohol has three -OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N.arrow_forward
- (a) Write the IUPAC name of the following molecule (b) Write the condensed structural formula of the following molecule OF THE MOLECULE OCTANE C8H18arrow_forwardAnswer this question:How to draw the line bond formula or lewis structure of a methyl ketone with the chemical formula C6H5C3H5O? (also taking into consideration the solubility test and chemical test results provided below) Based on the results of the solubility tests the compound is insoluble in water, 10% NaOH and 10% HCl but soluble in concentrated H2SO4. The functional group/class is identified to be Methyl Ketone, based on the results of the chemical tests on Table 2. CHEMICAL TEST OBSERVATIONS +(compound tested positive for the chemical reaction)/ otherwise (-) Molisch test turbid colorless solution - 2,4-DNP test formation of orange-yellow precipitates + Tollen’s test turbid colorless solution - Ninhydrin test clear pale-yellow solution - iodoform test clear pale-yellow solution +arrow_forwardDraw the structural formulas for the following compounds. Include all the bonds to hydrogen atoms. Be sure to answer both parts. (a) 1,4-dichloro-2-ethylbenzene: (b) 2-ethyl-1,3-dimethylbenzene:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License