
Concept explainers
Fill the blanks in the following table.
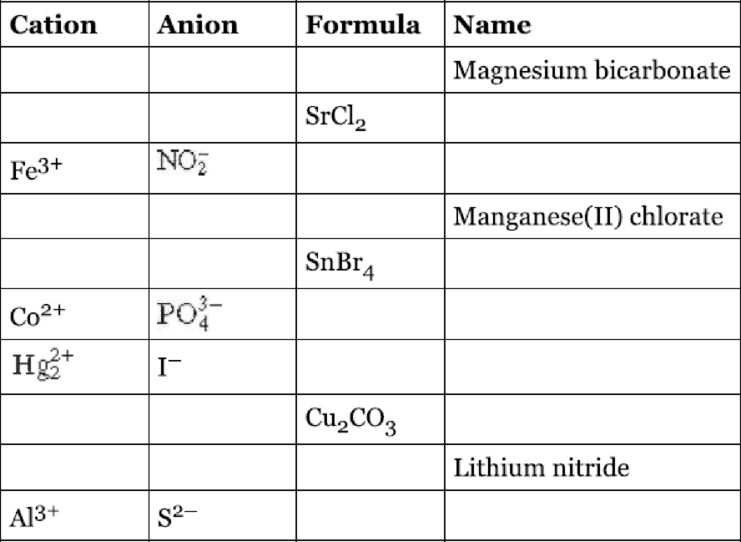
Interpretation:
The given table has to be filled.
Explanation of Solution
The given name is Magnesium bicarbonate. The cation and anion present in the compound are
The given formula is
The cation and anion present in the compound is
The given name is Manganese
The given formula is
The cation and anion present in the compound are
The cation and anion present in the compound are
The given formula is
The given name is Lithium nitride. The cation and anion present in the compound are
The cation and anion present in the compound are
The completed table is shown below,
| Cation | Anion | Formula | Name |
| Magnesium bicarbonate | |||
| Strontium chloride | |||
| Iron | |||
| Manganese | |||
| Tin | |||
| Cobalt | |||
| Mercury | |||
| Copper | |||
| Lithium nitride | |||
| Aluminum sulphide |
Want to see more full solutions like this?
Chapter 2 Solutions
Connect 1 Semester Access Card For Chemistry
- 7.101 Lead selenide nanocrystals may provide a breakthrough in the engineering of solar panels to be efficient enough to be an economical source of electricity. Selenium is generally considered a nonmetal while lead is considered a metal. Is this distinction enough to suggest that this compound should be ionic? Explain your answer.arrow_forwardA student in your chemistry class tells you that nitrogen-14 and nitrogen-15 are not isotopes because they have the same number of protons. How would you refute this statement?arrow_forwardTwo samples of different compounds of nitrogen and oxygen have the following compositions. Show that the compounds follow the law of multiple proportions. What is the ratio of oxygen in the two compounds for a fixed amount of nitrogen? Amount N Amount O Compound A 1.206 g 2.755 g Compound B 1.651 g 4.714 garrow_forward
- Which of the following is true about an individual atom? Explain. a. An individual atom should be considered to be a solid. b.An individual atom should be considered to be a liquid. c. An individual atom should be considered to be a gas. d. The state of the atom depends on which element it is. e. An individual atom cannot be considered to be a solid, liquid, or gas. Justify your choice, and for choices you did not pick, explain what is wrong with them.arrow_forwardMyocardial perfusion imaging (MPI) is the latest tool used in the detection of coronary artery disease. It uses thallium-201 chloride. How many neutrons are in (a) seventy atoms of thallium-201 ? (b) one picogram (11012g) of thallium-201?arrow_forwardFill in the following table:arrow_forward
- If an atom has 18 electrons, how many protons does it have?arrow_forwardA single molecule has a mass of 7.31 1023 g. Provide an example of a real molecule that can have this mass. Assume the elements that make up the molecule are made of light isotopes where the number of protons equals the number of neutrons in the nucleus of each element.arrow_forwardplease help me with filling in the table belowarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





