
ELEMENTS OF CHEM. REACTION ENGR
5th Edition
ISBN: 9780135486498
Author: Fogler
Publisher: INTER PEAR
expand_more
expand_more
format_list_bulleted
Textbook Question
thumb_up100%
Chapter 2, Problem 2.10P
The curve shown in Figure 2-1 is typical of a reaction carried out isothermally, and the curve shown in Figure P2-10B (sec Example 11-3) is typical of a gas-solid catalytic exothermic reaction carried out adiabatically).
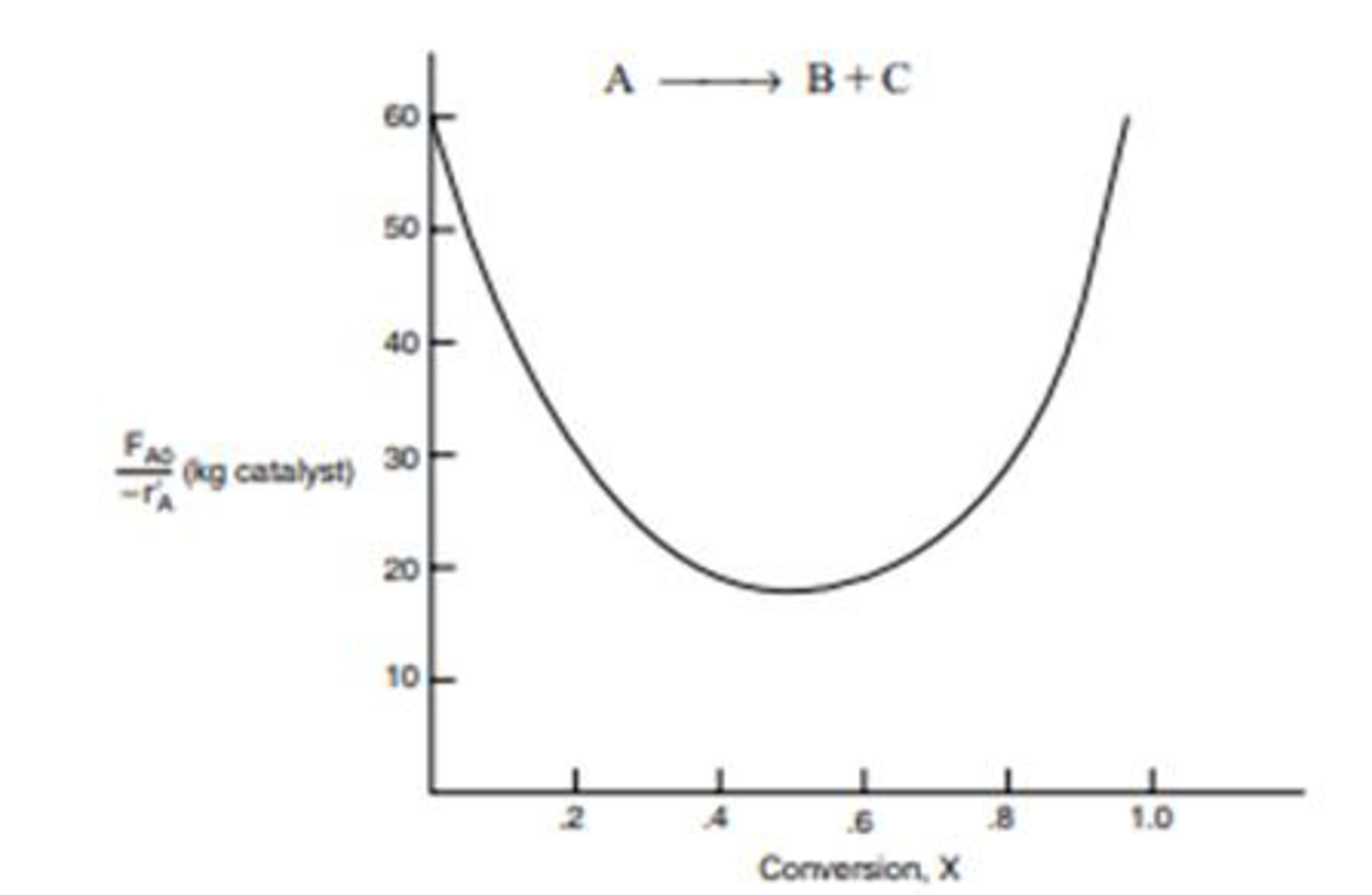
Figure P2-10B Levenspiel plot for an adiabatic exothermic heterogeneous reaction.
- (a) Assuming that you have a fluidized CSTR and a PBR containing equal weights of catalyst, how should they be arranged for this adiabatic reaction? Use the smallest amount of catalyst weight to achieve 80% conversion of A.
- (b) What is the catalyst weight necessary to achieve 80% conversion in a fluidized CSTR?
- (c) What fluidized CSTR weight is necessary to achieve 40% conversion?
- (d) What PBR weight is necessary to achieve 80% conversion?
- (e) What PBR weight is necessary to achieve 40% conversion?
- (f) Plot the rate of reaction and conversion as a function of PBR catalyst weight, W.
Additional information: FA0 = 2 mol/s.
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule05:37
Chapter 2 Solutions
ELEMENTS OF CHEM. REACTION ENGR
Additional Engineering Textbook Solutions
Find more solutions based on key concepts
T F The base class constructor is called after the derived class constructor.
Starting Out with C++ from Control Structures to Objects (9th Edition)
The size, shape, color and weight of an object are considered of the objects class.
Java How to Program, Early Objects (11th Edition) (Deitel: How to Program)
A cylinder having a mass of 250 kg is to be supported by the cord that wraps over the pipe. Determine the large...
Engineering Mechanics: Statics
The ball is kicked from point A with the initial velocity vA = 10 m/s. Determine the range R , and the speed wh...
Engineering Mechanics: Dynamics (14th Edition)
The ________ object is assumed to exist and it is not necessary to include it as an object when referring to it...
Web Development and Design Foundations with HTML5 (8th Edition)
Distinguish among data definition commands, data manipulation commands, and data control commands.
Modern Database Management (12th Edition)
Knowledge Booster
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The