
Where on the blank outline of the periodic table do elements that meet the following descriptions appear?
- (a) Elements with the valence-shell ground-state electron configuration ns2 np5
- (b) An element whose fourth shell contains two p electrons
- (c) An element with the ground-state electron configuration [Ar] 4s2 3d10 4p5
(a)
Interpretation:
Elements with the given valence shell electronic configuration has to be identified.
Explanation of Solution
Given valence shell electronic configuration,
Periodic table is given in figure 1.
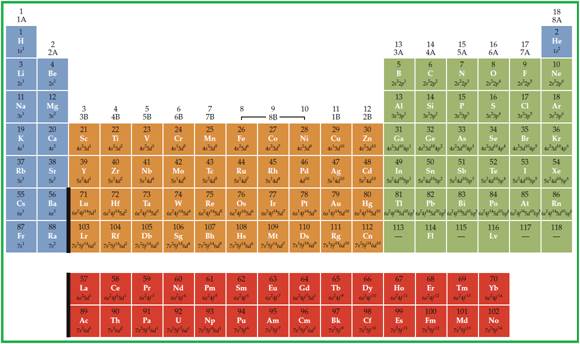
Figure 1
The elements with the given valence shell electronic configuration are highlighted (Red color) in the below periodic table
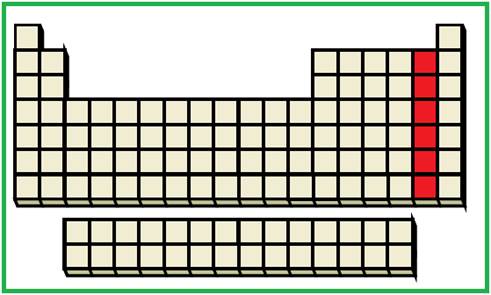
Figure 2
(b)
Interpretation:
An element with fourth shell containing two
Explanation of Solution
Given valence shell electronic configuration,
Periodic table is given in figure 1.

Figure 1
An element with fourth shell contains two
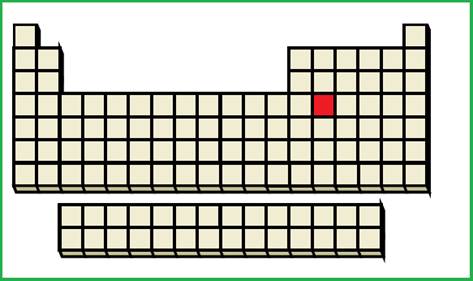
Figure 3
(c)
Interpretation:
An element with given ground-state electronic configuration has to be identified.
Explanation of Solution
Given ground-state electronic configuration,
Calculate the total number of electrons
Periodic table is given in figure 1.

Figure 1
An element with fourth shell contains two
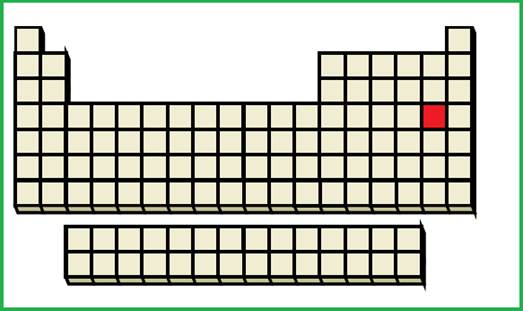
Figure 4
Want to see more full solutions like this?
Chapter 2 Solutions
General Chemistry: Atoms First-Solution Manual
- • identify an orbital (as 1s, 3p, etc.) from its quantum numbers, or vice versa.arrow_forwardIn what main group(s) of the periodic table do elements have the following number of half-filled p-orbitals in the outermost principal energy level? (a) 0 (b) 1(c) 2(d) 3arrow_forwardThe configuration of an element is given here. (a) What is the identity of the element? (b) In what group and period is the element found? (c) Is the element a nonmetal, a main group element, a transition metal, a lanthanide, or an actinide? (d) Is the element diamagnetic or paramagnetic? If paramagnetic, how many unpaired electrons are there? (e) Write a complete set of quantum numbers (n, , m, ms) for each of the valence electrons. (f) What is the configuration of the 2+ ion formed from this element? Is the ion diamagnetic or paramagnetic?arrow_forward

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





