
Concept explainers
(a)
Interpretation:
The isomeric structures of octane having five carbons in their principal chains are to be drawn. The naming for all the structures is to be stated.
Concept introduction:
The naming of the chemical compound is done using the parameters given by IUPAC. IUPAC stands for International Union of Pure and Applied Chemistry. This system of nomenclature is accepted worldwide. The IUPAC system provides the set of rules in order to do the naming of the chemical compounds.
Isomers are the chemical compounds which have same chemical formula but different arrangement of molecules.
Answer to Problem 2.26AP
The isomeric structures of octane having five carbons in their principal chains along with their names are drawn below as,
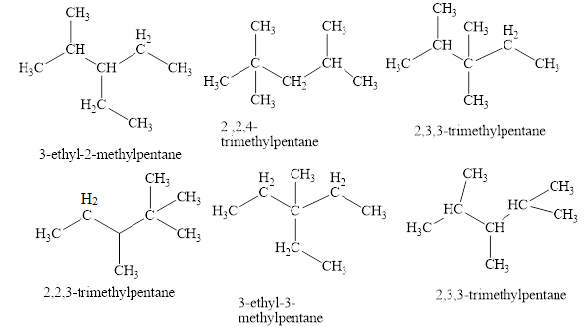
Explanation of Solution
The structure of octane can be drawn as shown in figure 1.
![]()
Figure 1
The isomers of the octane having five carbons in their principal chains can be drawn by rearranging the carbon atoms.
The first isomer is shown in figure 2 as,
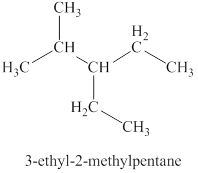
Figure 2
For the naming of the compound, the longest carbon chain is selected first. In the given compound the longest carbon chain has
The second isomer is shown in figure 3 as,
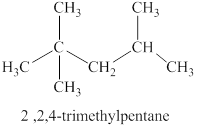
Figure 3
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The third isomer is shown in figure 4 as,
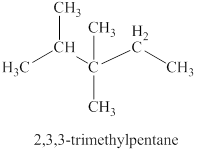
Figure 4
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fourth isomer is shown in figure 5 as,
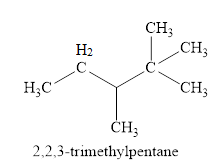
Figure 5
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fifth isomer is shown in figure 6 as,
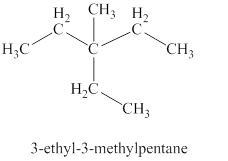
Figure 6
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The sixth isomer is shown in figure 7 as,
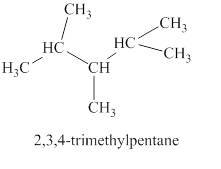
Figure 7
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The isomeric structures of octane having five carbons in their principal chains along with their names are shown in figure 2 to 7.
(b)
Interpretation:
The isomeric structures of octane having six carbons in their principal chains are to be drawn. The naming for all the structures is to be stated.
Concept introduction:
The naming of the chemical compound is done using the parameters given by IUPAC. IUPAC stands for International Union of Pure and Applied Chemistry. This system of nomenclature is accepted worldwide. The IUPAC system provides the set of rules in order to do the naming of the chemical compounds.
Isomers are the chemical compounds which have same chemical formula but different arrangement of molecules.
Answer to Problem 2.26AP
The isomeric structures of octane having six carbons in their principal chains along with their names are drawn below as,
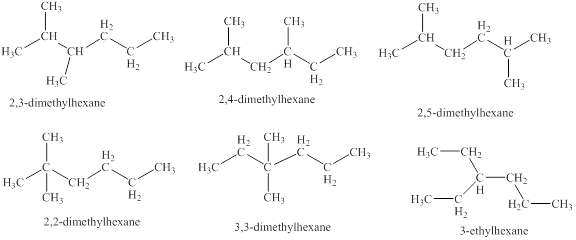
Explanation of Solution
The isomers of the octane having six carbons in their principal chains can be drawn by rearranging the carbon atoms.
The first isomer is shown in figure 8 as,
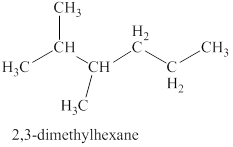
Figure 8
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The second isomer is shown in figure 9 as,
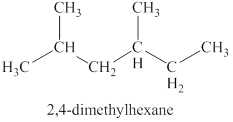
Figure 9
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The third isomer is shown in figure 10 as,
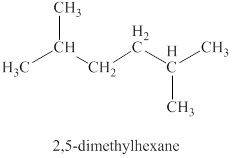
Figure 10
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fourth isomer is shown in figure 11 as,
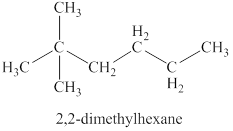
Figure 11
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fifth isomer is shown in figure 12 as,
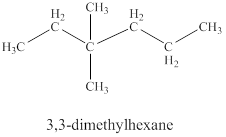
Figure 12
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The sixth isomer is shown in figure 13 as,
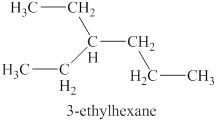
Figure 13
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest chain carbon has
The isomeric structures of octane having six carbons in their principal chains along with their names are shown in figure 8 to 13.
Want to see more full solutions like this?
Chapter 2 Solutions
Loose-leaf Version For Organic Chemistry
- There are eight structures with the formula C3H7NO in whichthe O is part of a carbonyl group. Draw the structures andidentify the functional groups in each.arrow_forwardGive the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardEncricle the functional groups in the followingf compounds, label each with the letters indicated and indentify the class in which the functional group may be foundarrow_forward
- Draw the structures and give the name of the 8 constitutional isomers with the molecular formula C9H12 that contain a benzene ring.arrow_forwardDraw the condensed structures of the compounds formedfrom (a) butanoic acid and methanol, (b) benzoic acid and2-propanol, (c) propanoic acid and dimethylamine. Namethe compound in each case.arrow_forwardhow can you tell there is isomers in butane and how can you label them with 2-methylpropanearrow_forward
- C12H17O2N ----> CH3I gives ( in structural formula ) ?arrow_forwardDraw all line structures of the constitutional isomers with the molecular formula C4H10O. What functional groups are possible?arrow_forwardDraw the skeletal isomers of chlorohexane, excluding those with branched carbon chains.arrow_forward
- Draw the structures of all monobromo derivatives of pentane, C5H11Br, which contain a 4-carbon chain.arrow_forwardDraw all possible structure(s) and give the IUPAC systematic name(s) of an alkane or cycloalkane with the formula C8H18 that has only primary hydrogen atoms...arrow_forwardThere are 11 structures (ignoring stereoisomerism) with the formula C4H8O that have no carbon branches. Draw the structures and identify the functional groups in each.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

