
ORGANIC CHEMISTRY W/BIO...-STUD.SOLN.
5th Edition
ISBN: 9781259920066
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.59P
Atenolol is a
the indicated
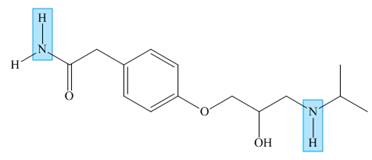
atenolol
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Atenolol is a β (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N—H bonds is more acidic? Explain your reasoning.
Is p-methylphenol more acidic than phenol? Why or why not?
Draw the structure of a constitutional isomer of compound B that fits each description. an isomer that is at least 105 times more acidic than B an isomer that is at least 105 times less acidic than B an isomer that is comparable in acidity to B
Chapter 2 Solutions
ORGANIC CHEMISTRY W/BIO...-STUD.SOLN.
Ch. 2 - a. Which compounds are Bronsted-Lowry acids:...Ch. 2 - a. Draw the conjugate acid of each base:...Ch. 2 - Label each statement as True or False.
a. is the...Ch. 2 - Label the acid and base, and the conjugate acid...Ch. 2 - Decide which compound is the acid and which is the...Ch. 2 - Draw the products formed from the acid-base...Ch. 2 - Which compound in each pair is the stronger acid?...Ch. 2 - Use a calculator when necessary to answer the...Ch. 2 - Rank the conjugate bases of each of group of acids...Ch. 2 - Problem-2.10 Considers two acids: (formic acid,)...
Ch. 2 - Prob. 2.11PCh. 2 - Draw the products of each reaction and determine...Ch. 2 - Prob. 2.13PCh. 2 - Without reference to a pKa table, decide which...Ch. 2 - Rank the labeled H atoms in the following compound...Ch. 2 - Which hydrogen in each molecule is most...Ch. 2 - Which hydrogen in pseudoephedrine, the nasal...Ch. 2 - Which compound in each pair of isomers is the...Ch. 2 - Which compound in each pair is the stronger acid?...Ch. 2 - Glycolic acid, HOCH2CO2H, is the simplest member...Ch. 2 - Explain the apparent paradox. HBr is a stronger...Ch. 2 - The CH bond in acetone, (CH3)2C=O, has a pKa of...Ch. 2 - Acetonitrile (CH3CN) has a pKa of 25, making it...Ch. 2 - For each pair of compounds: [1] Which indicated H...Ch. 2 - Rank the compounds in each group in order of...Ch. 2 - Which proton in each of the following drugs is...Ch. 2 - Which anion A or B is the stronger base? ABCh. 2 - Prob. 2.28PCh. 2 - Problem 2.29
Compounds like amphetamine that...Ch. 2 - Problem 2.30 Which species are Lewis bases?
a. b....Ch. 2 - Which species are Lewis acids?
a. b. c. d.
Ch. 2 - For each reaction, label the Lewis acid and base....Ch. 2 - Prob. 2.33PCh. 2 - Prob. 2.34PCh. 2 - Label the Lewis acid and base. Use curved arrow...Ch. 2 - 2.36 Propranolol is an antihypertensive agent—that...Ch. 2 - 2.37 Amphetamine is a powerful stimulant of the...Ch. 2 - 2.38 What is the conjugate acid of each base?
a....Ch. 2 - 2.39 What is the conjugate base of each acid?
a....Ch. 2 - 2.40 Draw the products formed from the acid-base...Ch. 2 - Draw the products formed from the acid-base...Ch. 2 - Draw the products of each proton transfer...Ch. 2 - Prob. 2.43PCh. 2 - Prob. 2.44PCh. 2 - What is Ka for each compound? Use a calculator...Ch. 2 - What is the pKa for each compound? a. b. c.Ch. 2 - Which of the following bases are strong enough to...Ch. 2 - Which compounds can be deprotonated by OH, so that...Ch. 2 - Draw the products of each reaction. Use the pKa...Ch. 2 - Rank the following compounds in order of...Ch. 2 - 2.51 Rank the following ions in order of...Ch. 2 - Prob. 2.52PCh. 2 - Prob. 2.53PCh. 2 - 2.54 The of three bonds is given below.
a. For...Ch. 2 - a. What is the conjugate acid of A? b. What is the...Ch. 2 - 2.56 Draw the structure of a constitutional isomer...Ch. 2 - 2.57 Many drugs are Bronsted-Lowry acids or...Ch. 2 - Dimethyl ether (CH3OCH3) and ethanol (CH3CH2OH)...Ch. 2 - 2.59 Atenolol is a (beta) blocker, a drug used to...Ch. 2 - 2.60 Use the principles in Section 2.5 to label...Ch. 2 - 2.61 Label the three most acidic hydrogen atoms in...Ch. 2 - Prob. 2.62PCh. 2 - 2.63 Classify each compound as a Lewis base, a...Ch. 2 - 2.64 Classify each species as a Lewis acid, a...Ch. 2 - Label the Lewis acid and Lewis base in each...Ch. 2 - 2.66 Draw the products of each Lewis acid-base...Ch. 2 - Prob. 2.67PCh. 2 - 2.68 Answer the following questions about the four...Ch. 2 - Prob. 2.69PCh. 2 - 2.70 Hydroxide can react as a Brønsted-Lowry base...Ch. 2 - 2.71 Answer the following questions about esmolol,...Ch. 2 - 2.72 DBU, is a base we will encounter in...Ch. 2 - 2.73 Molecules like acetamide can be protonated...Ch. 2 - Prob. 2.74PCh. 2 - Prob. 2.75PCh. 2 - 2.76 Write a stepwise reaction sequence using...Ch. 2 - Prob. 2.77PCh. 2 - 2.78 Which compound, M or N, is the stronger acid?...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict which member of each pair will be more acidic. Explain your answers. 2-chloropropan-1-ol or 3-chloropropan-1-olarrow_forwardRank the following bases in order of increasing basicity: LDA, cyclohexane magnesium chloride, NaOme, benzylamine, water.arrow_forwardRank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.arrow_forward
- Draw the structure of a constitutional isomer of compound B that fitseach description.a.) an isomer that is at least 105 times more acidic than Bb.) an isomer that is at least 105 times less acidic than Bc.) an isomer that is comparable in acidity to Barrow_forwardPropoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic. Explain why they are equally basic.arrow_forwardDraw the structure of a constitutional isomer of compound B that fits each description. a. an isomer that is at least 105 times more acidic than B b. an isomer that is at least 105 times less acidic than B c. an isomer that is comparable in acidity to Barrow_forward
- Rank the following compounds in their correct order of acidity. 1=Most acidic and 4=least acidic.arrow_forwardAlthough it was initially sold as a rat poison, warfarin is an effective anticoagulant used to prevent blood clots. Label the most acidic proton in warfarin, and explain why its pKa is comparable to the pKa of a carboxylic acid.arrow_forward1. Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2O2. Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds, and explain why the acidity increases so dramatically with substitution by nitro groups. CH4 CH3NO2 CH2(NO2)2 CH(NO2)3arrow_forward
- Which of the attached anions is the stronger base? Explain your choice.arrow_forwardArrange each group of compounds in order of increasing acidity.butyric acid, a@bromobutyric acid, b@bromobutyric acidarrow_forwardPredict which member of each pair will be more acidic and Explain 1) 2-Chloro-1-propanol or 3-chloro-1-propanol 2) p-nitro phenol or p-aminophenolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY