
Concept explainers
(a)
Interpretation: The product formed by the reaction of Lewis acid
Concept introduction: Lewis acid accepts an electron pair. It acts as an electrophile as it is electron deficient. Lewis base donates an electron pair. It acts as a nucleophile as it is electron loving.
Answer to Problem 2.67P
The product formed by the reaction of
Explanation of Solution
In the given reaction, Lewis base
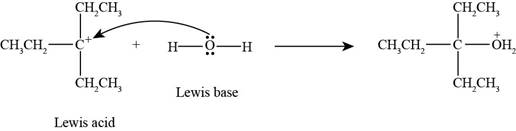
Figure 1
The product formed by the reaction of
(b)
Interpretation: The product formed by the reaction of Lewis acid
Concept introduction: Lewis acid accepts an electron pair. It acts as an electrophile as it is electron deficient. Lewis base donates an electron pair. It acts as a nucleophile as it is electron loving.
Answer to Problem 2.67P
The product formed by the reaction of
Explanation of Solution
In the given reaction, Lewis base
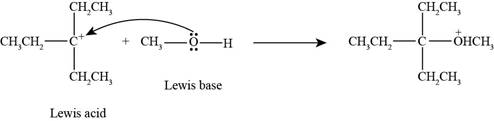
Figure 2
The product formed by the reaction of
(c)
Interpretation: The product formed by the reaction of Lewis acid
Concept introduction: Lewis acid accepts an electron pair. It acts as an electrophile as it is electron deficient. Lewis base donates an electron pair. It acts as a nucleophile as it is electron loving.
Answer to Problem 2.67P
The product formed by the reaction of
Explanation of Solution
In the given reaction, Lewis base
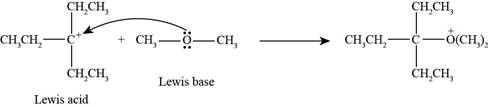
Figure 3
The product formed by the reaction of
Want to see more full solutions like this?
Chapter 2 Solutions
ORGANIC CHEM CONNECT ACCESS CARD
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forward1A. .Which of these are properties of acids? They are sour in taste. They turn red litmus paper blue. They dissolve many metals. Theyfeelslippery.A. I only. B. III only C. I and II. D. I and III. E. II, III, and IV 1B. Which of these is/are isomers of butane? A. propane B. propaneC. 2-metylpropane D. 2-methylbutane E. pentane 1c. Which is the oxidizing agent in, 2CO + 2NO → 2CO2 + N2? CO NO CO2 N2 CO and NO 1d. Which is the reducing agent in, Cu (s) + 2Ag+ (aq) → Cu2+ (aq) + 2Ag (s)? Cu2+ Ag Cu Ag+ H2O, since the reaction occursarrow_forwardDraw structures sudan orange with HCL/ water / NAOH.arrow_forward
- Assume for the purposes of this problem that to be an alcohol (-ol) or an amine (-amine), the hydroxyl or amino group must be bonded to a tetrahedral (sp3 hybridized) carbon atom. Write the structural formula of a compound with an unbranched chain of four carbon atoms that is an: Q. Alkynoic acidarrow_forwardWhere does the linkage between the -COOH group on one end of one molecule and the -NH2 group at the end of another molecule occur in?arrow_forwardFormaldehyde, H2C=O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no carbonyl groups. Only one monobromo derivative (C3H5BrO3) of trioxane is possible. Propose a structure for trioxane.arrow_forward
- Does alcohol, ketones, and aldehydes considered acidic or a basic?arrow_forwardExplain why acetic acid, CH3CO2H, Contains two distinct types of Carbon oxygen Bonds, Whereas the acetate ion, formed by loss of hydrogen ion from acetic acid, Only contains one type of carbon oxygen bondarrow_forwardHow can I attach an amine group (-NH2) to a carboxyl group (-COOH)? What are the experimental conditions, such as temperature, atmosphere, pH etc?arrow_forward
- Assume for the purposes of this problem that to be an alcohol (-ol) or an amine (-amine), the hydroxyl or amino group must be bonded to a tetrahedral (sp3 hybridized) carbon atom. Write the structural formula of a compound with an unbranched chain of four carbon atoms that is an: Q. Alkenolarrow_forwarddraw 2 isomers tertiary amine with the formula C5H13Narrow_forwardHCO3-arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





