
LAB MANUAL A&P & MOD MSTG/ET SA ACC PK
6th Edition
ISBN: 9780134838007
Author: Marieb
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 4SAQ
Consider the following information about three atoms:
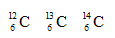
(a) How are they similar to one another? (b) How do they differ from one another? (c) What are the members of such a group of atoms called? (d) Using the planetary model, draw the atomic configuration of 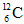 showing the relative position and numbers of its subatomic particles.
showing the relative position and numbers of its subatomic particles.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Where within an atom are the three types of subatomic particles located?
Which of the following levels in the shell model of an atom can hold up to two electrons?
A. The second shell
B. The first shell
C. The third shell
Look up antimony in the periodic table 1Z = 512. How many covalent bonds would you expect it to form? Based on this information, which of the following antimony compounds is covalent and which is ionic: SbCl3 or SbCl5?
Chapter 2 Solutions
LAB MANUAL A&P & MOD MSTG/ET SA ACC PK
Ch. 2.1 - What form of energy is found in the food we eat?Ch. 2.1 - What form of energy is used to transmit messages...Ch. 2.1 - What type of energy is available when we are...Ch. 2.2 - What two elements besides H and N make up the bulk...Ch. 2.2 - An element has a mass of 207 and has 125 neutrons...Ch. 2.2 - How do the terms atomic mass and atomic weight...Ch. 2.3 - What is the meaning of the term molecule?Ch. 2.3 - Why is sodium chloride (NaCl) considered a...Ch. 2.3 - Blood contains a liquid component and living...Ch. 2.4 - What kinds of bonds form between water molecules?
Ch. 2.4 - Oxygen (8O) and argon (18A) are both gases. Oxygen...Ch. 2.4 - Assume imaginary compound XY has a polar covalent...Ch. 2.5 - Which reaction type-synthesis, decomposition, or...Ch. 2.5 - Why are many reactions that occur in living...Ch. 2.5 - What specific name is given to decomposition...Ch. 2.6 - Salts are electrolytes. What does that mean?Ch. 2.6 - Which ion is responsible for increased acidity?Ch. 2.6 - To minimize the sharp pH shift that occurs when a...Ch. 2.6 - Prob. 19CYUCh. 2.7 - Prob. 20CYUCh. 2.8 - What are the monomers of carbohydrates called?...Ch. 2.8 - What is the animal form of stored carbohydrate...Ch. 2.9 - Prob. 23CYUCh. 2.10 - What does the name amino acid tell you about the...Ch. 2.10 - What is the primary structure of proteins?Ch. 2.10 - What are the two types of secondary structure in...Ch. 2.10 - How do enzymes reduce the amount of activation...Ch. 2.11 - How do DNA and RNA differ in the bases and sugars...Ch. 2.11 - What are two important roles of DNA?Ch. 2.12 - Glucose is an energy-rich molecule. So why do body...Ch. 2.12 - What change occurs in ATP when it releases energy?Ch. 2 - Which of the following forms of energy is the...Ch. 2 - All of the following are examples of the four...Ch. 2 - The mass number of an atom is (a) equal to the...Ch. 2 - A deficiency in this element can be expected to...Ch. 2 - Which set of terms best describes a proton? (a)...Ch. 2 - The subatomic particles responsible for the...Ch. 2 - Prob. 7MCCh. 2 - Which of the following does not describe a...Ch. 2 - In a beaker of water, the water-water bonds can...Ch. 2 - When a pair of electrons is shared between two...Ch. 2 - Molecules formed when electrons are shared...Ch. 2 - Which of the following covalently bonded molecules...Ch. 2 - Prob. 13MCCh. 2 - Factors that accelerate the rate of chemical...Ch. 2 - Prob. 15MCCh. 2 - Waters importance to living systems reflects (a)...Ch. 2 - Acids (a) release hydroxyl ions when dissolved in...Ch. 2 - Prob. 18MCCh. 2 - Prob. 19MCCh. 2 - A chemical has an amine group and an organic acid...Ch. 2 - Prob. 21MCCh. 2 - Enzymes are organic catalysts that (a) alter the...Ch. 2 - Define or describe energy, and explain the...Ch. 2 - Some energy is lost in energy energy conversion....Ch. 2 - Provide the atomic symbol for each of the...Ch. 2 - Consider the following information about three...Ch. 2 - How many moles of aspirin, C9H8O4, are in a bottle...Ch. 2 - Given the following types of atoms, decide which...Ch. 2 - What are hydrogen bonds and how are they important...Ch. 2 - Prob. 8SAQCh. 2 - Differentiate clearly between primary, secondary,...Ch. 2 - Prob. 10SAQCh. 2 - Describe the mechanism of enzyme action.Ch. 2 - Explain why, if you pour water into a glass very...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Magnesium has an atomic number of 12. Which of the following statements is true of a neutral magnesium atom? a. It has 12 protons, 12 electrons, and 12 neutrons. b. It has 12 protons, 12 electrons, and six neutrons. c. It has six protons, six electrons, and no neutrons. d. It has six protons, six electrons, and six neutrons.arrow_forwardHow many electrons are in the outer shell of each of the following atoms?arrow_forwardwhat are the parts of an atom located? What is the structure? what are these subatomic particles give to an atom?arrow_forward
- Which electron shell is the valence shell of an atom, and what is its significance?arrow_forwardDefine the atomic mass of an atom. What are two things that make up an atom?arrow_forwardHow does the atomic structure or composition of the versions of sodium in question C above differ from a typical sodium atom, with its atomic mass of 23?arrow_forward
- [ Ionic cluster , covalent cluster , metallic cluster , hydrogen cluster , van der Waals cluster ] What is the definition of each of the following with image an example of it ??arrow_forwardWhen two atoms share a pair of valence electrons with different levels of electronegativity, what type of bond is this?arrow_forwardWhat is the total number of atoms in the molecule HNO3?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning

Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license