
Concept explainers
(a)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(a)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 1.
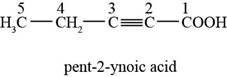
Figure 1
The structure comprises chain of five carbon atoms along with carboxylic acid at first and triple bond in between second and third carbon atoms. According to
Therefore, the
(b)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(b)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 2.
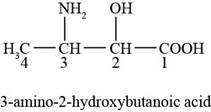
Figure 2
The structure comprises chain of four carbon atoms along with carboxylic acid group, hydroxyl group and amino group at first, second, and third carbon atoms respectively. Carboxylic acid group gets higher priority during the naming of structure because it is present at first carbon atom. According to
Therefore, the
(c)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(c)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 3.
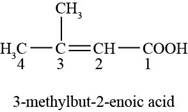
Figure 3
The structure comprises chain of four carbon atoms along with carboxylic acid group and methyl group at first and third carbon atoms respectively. The double bond is present in between the second and third carbon atoms. Carboxylic acid group gets higher priority during the naming of structure because it is present at first carbon atom. According to
Therefore, the
(d)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(d)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 4.
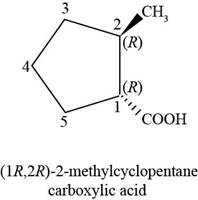
Figure 4
The structure comprises cyclic ring of five carbon atoms along with one group of carboxylic acids at first carbon atom and one methyl group at second carbon atom. The carboxylic acid group is present below the plane, whereas methyl group is present at above the plane. According to
Therefore, the
(e)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(e)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 5.
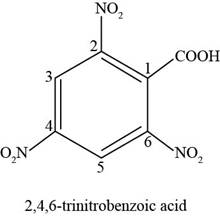
Figure 5
The structure comprises cyclic ring of six carbon atoms along with one carboxylic acid group at first position and three nitro groups at second, fourth and sixth carbon atoms. According to
Therefore, the
(f)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(f)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 6.
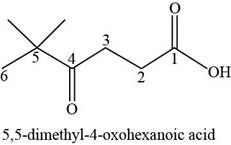
Figure 6
The structure comprises parent chain of six carbon atoms along with carboxylic acid group, carbonyl group, and two methyl groups at first, fourth and fifth carbon atoms respectively. According to
Therefore, the
Want to see more full solutions like this?
Chapter 20 Solutions
Student's Solutions Manual for Organic Chemistry
- Draw 3 structural isomers of isoamyl alcohols. Two must be an alcohol and one must be an ether.arrow_forwardGive iupac name for this diol CH3CH(OH)(CH2)4CH(OH)C(CH3)3arrow_forwardConsider the following test samples: A. heptane D. toluene B. hexane E. benzene C. hexene Which of the test samples will react with conc. H2SO4 ?arrow_forward
- Identify the organic compounds CH3CH(OH)CH2CHO and CHO in the equation:CH3CH(OH)CH2CHO ---> CH3CH + CHO +H20 a. butanol, cis/trans butanol b. butanoic acid, butanoic acid c. carbonyl aldehyde, cis or trans isomer d. butanol, butaldehydearrow_forward1. Write down the structural formulas for three isomeric compounds containing the carbonyl group and having the molecular formula C4H8O. Then give it the IUPAC name. 3. What is the product of the following reaction?arrow_forwardGive the IUPAC name for this organic compound: CH3-CH2-CHBr-CH=CH-CHBr-CH3 3,6-dibromo hept-4-ene 2,5-dibromo-4-heptene 2,5-dibromo hept-3-ene 3.6-dibromo-3- heptenearrow_forward
- Explain NaBH4 reductions used in organic synthesis ?arrow_forwardIsooctane is the common name of the isomer of C8H18 used as the standard of 100 for the gasoline octane rating: (a) What is the IUPAC name for the compound? (b) Name the other isomers that contain a five-carbon chain with three methyl substituents.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





