
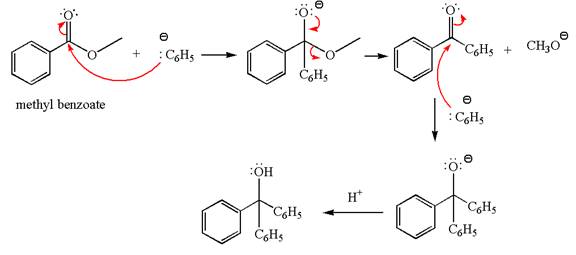
Concept explainers
(a)
Interpretation:
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be converted to a primary alcohol by reacting with
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
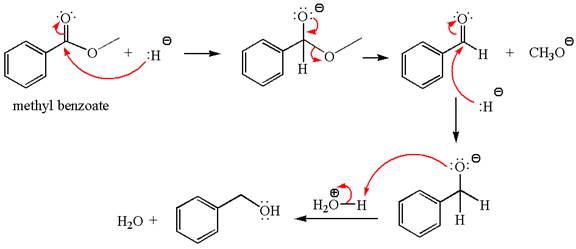
Explanation of Solution
The equation for the reaction of methyl benzoate with
Methyl benzoate is an ester, which, on reaction with
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(b)
Interpretation:
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be converted to a tertiary alcohol by reacting it with
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
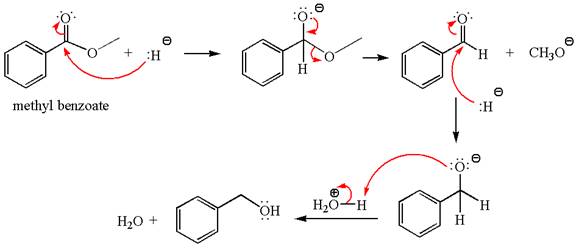
Explanation of Solution
The equation for the reaction of methyl benzoate with
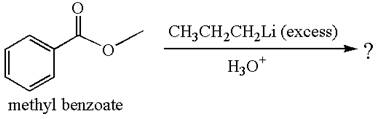
Methyl benzoate is an ester, which, on reaction with
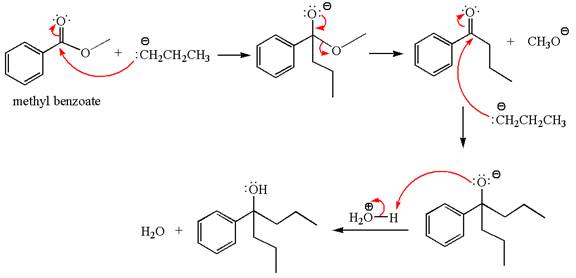
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(c)
Interpretation:
Whether methyl benzoate can react with
Concept introduction:
The reagent
Answer to Problem 20.46P
Mmethyl benzoate cannot react with
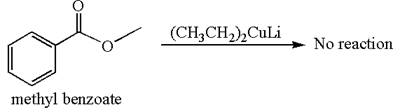
Explanation of Solution
The equation for the reaction of methyl benzoate with
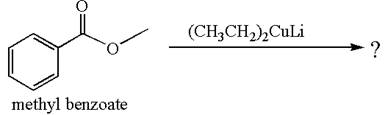
Methyl benzoate is an ester and
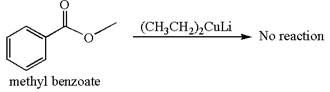
It is determined that no reaction occurs based on the reactivity of
(d)
Interpretation:
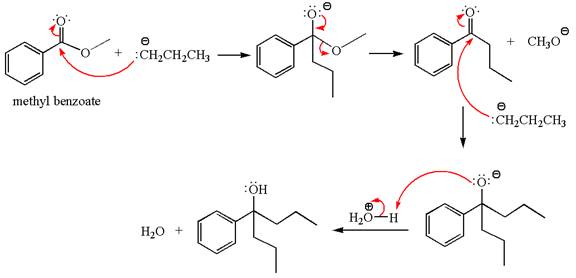
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be converted to a tertiary alcohol by reacting it with
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
Explanation of Solution
The equation for the reaction of methyl benzoate with
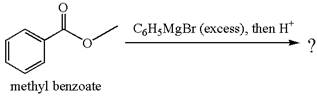
Methyl benzoate is an ester, which, on reaction with

The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(e)
Interpretation:
The product with detailed mechanism for the reaction between methyl benzoate and
Concept introduction:
An ester can be reduced to aldehyde without reducing it further to alcohol using a specific reagent such as
Answer to Problem 20.46P
The product with detailed mechanism for the reaction between methyl benzoate and
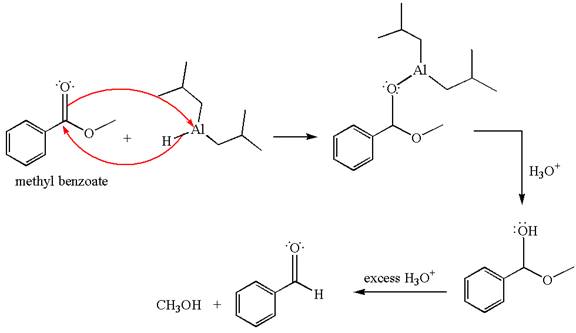
Explanation of Solution
The equation for the reaction of methyl benzoate with
Methyl benzoate is an ester, which, on reaction with
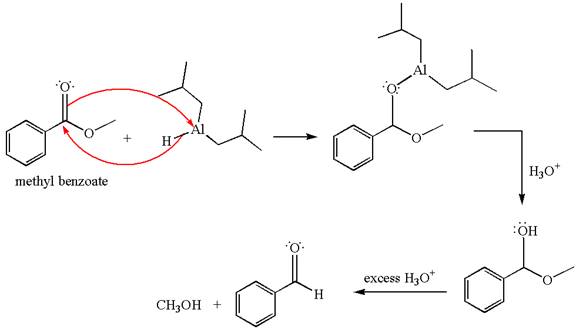
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
Want to see more full solutions like this?
Chapter 20 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- An ether can be prepared from an alkene and an alcohol by electrophilic addition, as shown in the reaction below. H+ CH₂CH₂OH Complete the mechanism for this reaction by adding curved arrows and products. Add steps as necessary, and be sure to include lone pairs and charges where relevant.arrow_forwardDraw a multi-step mechanism for this reaction and predict the structure of the organicproduct. reaction is in the imagearrow_forward4. identify the structure of A and B in the following synthetic scheme: Write out complete reactions for each step, showing the structure of all reactants and products. (a) cyclohexanol +Na2Cr2O7/H2SO4, H2O -------> A (b) A + Et2NH/H2SO4 ----->Barrow_forward
- Write the mechanism of the following reactions and find the product formed. c) CH,OH ?arrow_forwardAn electron-deficient carbon atom reacts with a nucleophile, symbolized as: Nu−. Define this ?arrow_forwardWhich of the following chemical equations depicts an alkylation reaction? C6H6() + CH3Cl() → C6H5CH3() + HCl(g) 2 CH3OH() + 3 O2(g) → 2 CO2(g) + 4 H2O() C6H12() → C6H10() + H2(g) CH2ClCH2Cl(g) + H2(g) → CH3CH3(g) + Cl2(g) CHClCHCl(g) → CH2ClCH2Cl(g)arrow_forward
- (a) What functional group is undergoing a transformation in the reaction? (b) What functional group is it being transformed into (in the final product)?arrow_forward11) Synthesize 2-methyl-3-hydroxycyclohexone from cyclohexone, methyl iodide, and inorganic precursors.arrow_forward3) Beginning from acetylene and any alkyl halide needed, how would you prepare the following compound, there may be more than one step. HC=CH CH;CH,CH,CH,CH,CH,CH;arrow_forward
