
Concept explainers
Synthesize each compound from the given starting material. You may use any other required inorganic reagents.
a. 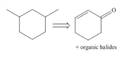 c.
c. 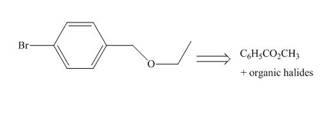
b. 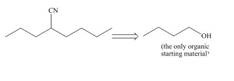 d.
d.
e.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry - With Access (Custom)
- taken in order to gor the product Please explain the mechanisms/stepsarrow_forwardB. Draw structures for the products in the following reactions. my NH₂ NH₂ 1. LIAIH4 2. H₂O* xs EtBr 1. Mel 2. Ag₂O, H,O, heat acetone, formaldehyde cat. HCIarrow_forward9. What is the major organic product obtained from the following reaction? 1. BH3 2. H₂O₂, NaOHarrow_forward
- Which reagent(s) convert a carbonyl group (C=O) into a methylene group (CH2)? O A. Zn / HCI O B. LIAIH4 / Et20 O C. NABH4 / MeOH D. Na in liq. NH3arrow_forwardDraw the structure of a dihalide that could be used to prepare each alkyne. There may be more than one possible dihalide. CH3 b. CH₂-C-C=CH CH₂ a. CH₂C=CCH₂ C. -C=C-arrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forward
- Does the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forwardComplete each hydrogenation reaction. catalyst a. CH2=CH-CH3 + H2 catalyst b. CH3-CH-CH=CH2 + H2 ČH3 catalyst c. с. CH, —CH—С—СH, + H, ČH; ČH;arrow_forwardH. An ester is produced by the reaction of an alcohol with a carboxylic acid. For each ester below, underline the alcohol part and circle the carboxylic acid part. Then, name each ester. CHỊCH,CH,CH,CH,-O-C-CH,CH,CH, H CH₂ CH₂ CH 1 CH3arrow_forward
- Complete the following halogenation reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cty-CH-0H b) CH3 CH-CH-CH PBY3 c) PE 7. Name the following phenols. d) a) b) OH c) OH Br OH CH2 HO CI Br 8. Name the following ethers. b) a) CHy-0-C-CHs CHy-0-CH3 c) d) Cy-CH-0 talamc) Earrow_forward16. Identify each compound as an a cohol, a phenol, or an ether. Classify any alcohols as primary (1"), secondary (2), or tertiary (3"). a. CH,CH,CH,OH CH,CHCH, b. CHO C. CH CHOCH, CH, d.arrow_forward3. Ethanoic acid (vinegar) when diluted to low concentrations by water can be prepared from ethene by a) reduction with H2, followed by reaction with a strong oxidizer b) addition of HCL followed by reaction with H20 c) addition of H20 followed by reaction with a strong oxidizer d) addition of Br2 e) followed by reduction with H2arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
