
Concept explainers
Reaction of butanenitrile (
(
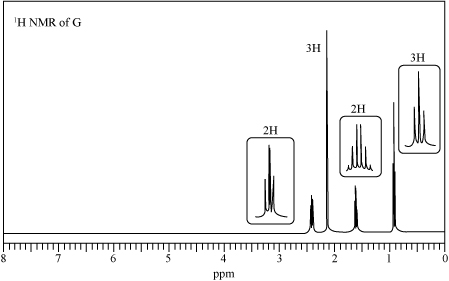
Trending nowThis is a popular solution!

Chapter 20 Solutions
ORGANIC CHEMISTRY(LL)W/ACCESS>CUSTOM<
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forward3-Chlorocyclopropene, on treatment with AgBF4, gives a precipitate of AgCl and a stable solution of a product that shows a single 1H NMR absorption at 11.04 δ. What is a likely structure for the products, and what is its relation to HĂ¼ckel’s rule?arrow_forwardA solution of acetone [(CH3)2C=O] in ethanol (CH3CH2OH) in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm−1, and the 1H NMR spectrum is given here. What is the structure of the product?arrow_forward
- Assume a compound with the formula C4H8O. a) How many double bonds and/or rings does your compound contain? b) If your compound shows an infrared absorption peak at 1715 cm-1, what functional group does it have? c) If your compound shows a single 1H NMR absorption peak at 2.1 δ, what is its structure?arrow_forwardTreatment of 2-methylpropanenitrile [(CH3)2CHCN] with CH3CH2CH2MgBr, followed by aqueous acid, affords compound V, which has molecular formula C7H14O. V has a strong absorption in its IR spectrum at 1713 cm−1, and gives the following 1H NMR data: 0.91 (triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and 2.60 (septet, 1 H) ppm. What is the structure of V? We will learn about this reaction in Chapter 20.arrow_forward19. A compound with molecular formula C6H12O2 exhibits two singlets in its 1H NMR spectrum, at d 1.4 (9H) and d 2.0 (3H). Its IR spectrum shows a strong absorption band near 1740 cm-1. What is the structure for this compound? Show the correlation of the spectra with the molecular structure.arrow_forward
- A chiral compound Y has a strong absorption at 2970−2840 cm−1 in its IR spectrum and gives the following mass spectrum. Propose a structure for Y.arrow_forwardTreatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

