
Concept explainers
Practice Problem 21.12
What products would form when each of the following compounds is treated with
(a)
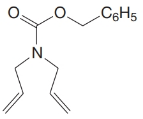
(b)
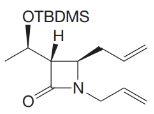
(c)
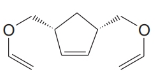
(d)
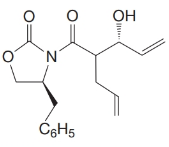
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
ORGANIC CHEMISTRY-ETEXT REG ACCESS
Additional Science Textbook Solutions
Living by Chemistry
Inorganic Chemistry
Chemistry: The Central Science (14th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
Chemistry: A Molecular Approach
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
- [ 21] What is major product of treating following compound with hot concentrated potassium permanganate followed by treatment with dilute HCl ? .COOH .COOH СОН СООН ОН Он COOH `COH `COOH ČOOH (a) (b ) (c) (d ) (e )arrow_forwardDraw a structural formula for the product formed by treating butanal with each reagent. (a) LiA1H4LiA1H4 followed by H2OH2O (b) NaBH4NaBH4 in CH3OH/H2O (c) H2/Pt (d) Ag(NH3)2+in NH3/H2O (e) H2CrO4, heat (f) HOCH2CH2OH,HClarrow_forward19.54 Predict the major product(s) from the treatment of acetone with the following: (d) [H], (CH3)2NH, (-H2O) (e) [H], NH2NH2, (-H2O) (f) [H], NH2OH, (-H2O) (g) NaBH4, MeOH (h) RCO3H (i) HCN, KCN (j) EtMgBr followed by H₂O (k) (C6H5)3P=CHCH2CH3 (1) LIAIH, followed by H2Oarrow_forward
- Quinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure. One step in the synthesis of quinapril involvesreaction of the racemic alkyl bromide A with a single enantiomer of theamino ester B. Given the structure of quinapril, which one of these two products isneeded to synthesize the drug?arrow_forward19.73 Propose an efficient synthesis for each of the following transformations: (d) OH -CN 0-0 Q-8" (e)arrow_forward32 Draw a structural formula for the product formed by treatment of butanal with each set of reagents. (a) H₂/transition metal catalyst (b) NaBH, then H₂O + (c) Ag(NH₂), (Tollens' reagent) (d) K₂Cr₂O/H₂SO4 (darrow_forward
- Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed to synthesize the drug?arrow_forward(c) Treating lactone B with two equivalents of phenylmagnesium bromide, followed by hydrolysis in aqueous acid, gives a compound with the molecular formula C18H22O2, as shown below. Propose a structural formula for this compound. 1. PhMgBr (2 eq.) 2. H'/H>O C18H2202 bj 1o 14 10I f6 40 & 3 4 00 (8)arrow_forwardWhen phenyl 3,4-dimethylbenzoate undergoes electrophilic aromatic substitution, one ring reacts much more readily than the other. (a) Which one is it? Encircle the ring (b) Explain your answer. (c) Draw the resulting product/s when it undergoes bromination reaction. Product (c) (a) Explanation (b)arrow_forward
- Predict the products when each compound undergo Ozonolysis (a) (b) (c) , (d) (e)arrow_forwardWhat steps are needed to convert benzene into p-isobutylacetophenone, a synthetic intermediate used in the synthesis of the anti-inammatory agent ibuprofen.arrow_forwardLinalool (the Chapter 9 opening molecule) and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can’t linalool be prepared by a similar pathway?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

