
ORGANIC CHEMISTRY-OWL V2 ACCESS
8th Edition
ISBN: 9781305582422
Author: Brown
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
thumb_up100%
Chapter 21, Problem 21.26P
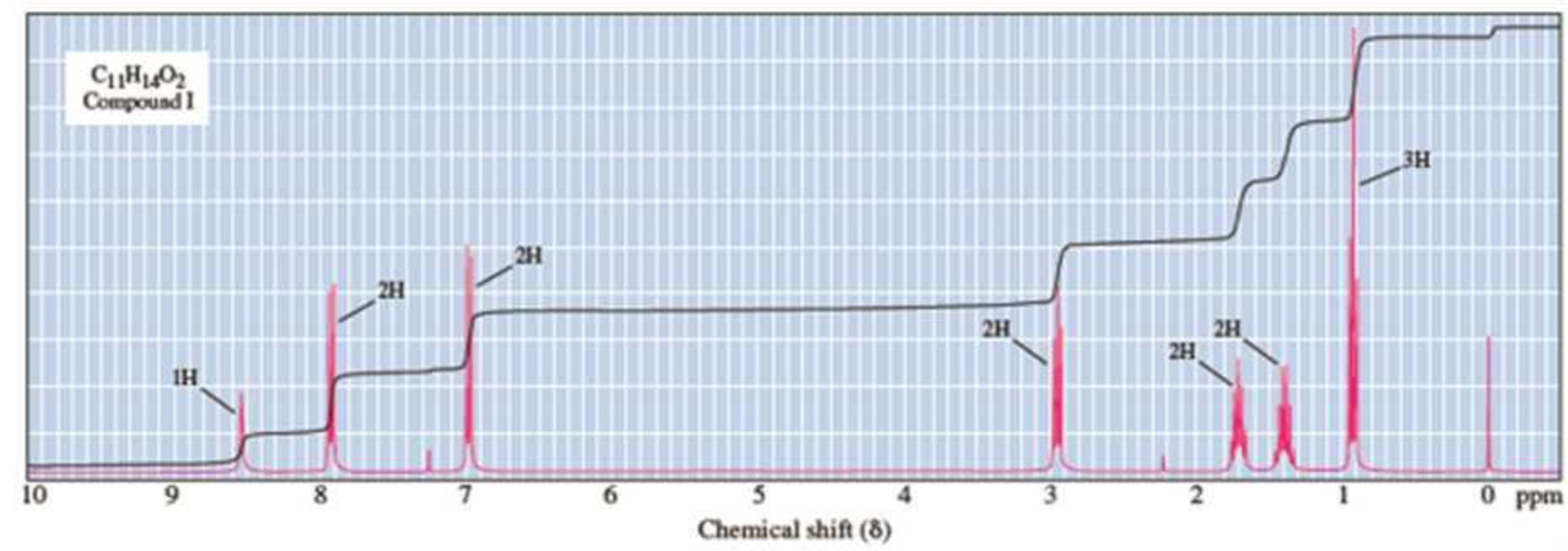
Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)
Compound A has molecular formula C5H10O. It shows three signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.1 ppm, a singlet of integral 3 at 2.14 ppm, and a quintet of integral 1 at 2.58 ppm. Suggest a structure for A and explain your reasoning.
A and B are isomeric dicarbonyl compounds of the molecular formula C5H&O2. The 'H NMR spectrum of A contains a singlet at 2.05 ppm and another
singlet at 5.40 ppm. The 'H NMR spectrum of B contains three signals: a singlet at 2.3 ppm, a triplet at 1.10 ppm and a quartet at 2.70 ppm. Suggest
structures for A and B and draw them in their respective boxes below.
1st attempt
Chapter 21 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
Ch. 21.2 - Construct a Frost circle for a planar...Ch. 21.2 - Which compound gives a signal in the 1H-NMR...Ch. 21.2 - Prob. 21.3PCh. 21.2 - Prob. 21.4PCh. 21.3 - Prob. 21.5PCh. 21.4 - Arrange these compounds in order of increasing...Ch. 21.4 - Prob. AQCh. 21.4 - Prob. BQCh. 21.4 - Prob. CQCh. 21.5 - Prob. 21.7P
Ch. 21 - Name the following compounds and ions.Ch. 21 - Prob. 21.9PCh. 21 - Draw a structural formula for each compound. (a)...Ch. 21 - Molecules of 6,6-dinitrobiphenyl-2,2-dicarboxylic...Ch. 21 - Following each name is the number of Kekul...Ch. 21 - Prob. 21.13PCh. 21 - Prob. 21.14PCh. 21 - Prob. 21.15PCh. 21 - Which of the molecules and ions given in Problem...Ch. 21 - Prob. 21.17PCh. 21 - Naphthalene and azulene are constitutional isomers...Ch. 21 - Prob. 21.19PCh. 21 - Prob. 21.20PCh. 21 - Following are IR and 1H-NMR spectra of compound D....Ch. 21 - Compound E (C8H10O2) is a neutral solid. Its mass...Ch. 21 - Following are 1H-NMR and 13C-NMR spectral data for...Ch. 21 - Following are 1H-NMR and 13C-NMR spectral data for...Ch. 21 - Compound H (C8H6O3) gives a precipitate when...Ch. 21 - Compound I (C11H14O2) is insoluble in water,...Ch. 21 - Propose a structural formula for compound J...Ch. 21 - Propose a structural formula for the analgesic...Ch. 21 - Prob. 21.29PCh. 21 - Prob. 21.30PCh. 21 - Given here are 1H-NMR and 13C-NMR spectral data...Ch. 21 - Prob. 21.32PCh. 21 - Prob. 21.33PCh. 21 - Prob. 21.34PCh. 21 - Arrange the molecules and ions in each set in...Ch. 21 - Prob. 21.36PCh. 21 - Prob. 21.37PCh. 21 - From each pair, select the stronger base.Ch. 21 - Prob. 21.39PCh. 21 - Prob. 21.40PCh. 21 - Prob. 21.41PCh. 21 - Prob. 21.42PCh. 21 - Following is an equation for iodination of...Ch. 21 - Prob. 21.44PCh. 21 - Prob. 21.45PCh. 21 - Prob. 21.46PCh. 21 - When warmed in dilute sulfuric acid,...Ch. 21 - In the chemical synthesis of DNA and RNA, hydroxyl...Ch. 21 - Prob. 21.49PCh. 21 - Write the products of the following sequences of...Ch. 21 - Prob. 21.51PCh. 21 - Show how to convert 1-phenylpropane into the...Ch. 21 - Prob. 21.53PCh. 21 - Cromolyn sodium, developed in the 1960s, has been...Ch. 21 - Prob. 21.55PCh. 21 - Prob. 21.56PCh. 21 - Prob. 21.57PCh. 21 - Prob. 21.58PCh. 21 - Prob. 21.59PCh. 21 - Prob. 21.60PCh. 21 - Prob. 21.61PCh. 21 - Prob. 21.62PCh. 21 - Prob. 21.63PCh. 21 - Following is a synthesis for toremifene, a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compound A with molecular formula C6H10 has two peaks in its 1H NMR spectrum, both of which are singlets (with ratio 9 : 1). Compound A reacts with an acidic aqueous solution containing mercuric sulfate to form compound B, which gives a positive iodoform test and has an 1H NMR spectrum that shows two singlets (with ratio 3 : 1). Identify A and B.arrow_forwardCompound C has the molecular formula C5H8O. The IR, 1H, 13C, and DEPT NMR spectra of this compound are shown below. (a) Calculate the double bond equivalent of compound C and briefly explain what the values obtains represents. Interpret the IR spectrum. (b) Based on the information provided, determine the structure of compound D.arrow_forwardTreatment of compound C (molecular formula C4H8O) with C6H5MgBr, followed by H2O, affords compound D (molecular formula C10H14O). Compound D has a strong peak in its IR spectrum at 3600–3200 cm−1. The 1H NMR spectral data of C and D are given. What are the structures of C and D? Compound C signals at 1.3 (singlet, 6 H) and 2.4 (singlet, 2 H) ppm Compound D signals at 1.2 (singlet, 6 H), 1.6 (singlet, 1 H), 2.7 (singlet, 2 H), and 7.2 (multiplet, 5 H) ppmarrow_forward
- Compound A is treated with a mixture of nitric and sulfuric acids to generate Compound B. The 1H-NMR spectrum of B shows two singlets, one at 2.52 pm and one at 8.13 ppm. The 13C-NMR spectrum of B shows five signals. The mass spectrum of B shows a peak at m/z = 260 and another peak at m/z = 262; the relative height of the two peaks is 1:1 respectively. - Identify compound B, explaining your reasoningarrow_forwardThe 1H-NMR spectrum of Compound D of molecular formula C10H12O shows three singlets – δ 2.20 (6H, s), 4.86 (4H), 7.10 (2H) ppm. Its 13C-NMR spectrum has five signals – 20, 74, 127, 135, 146 ppm. Suggest a structure for this compound.arrow_forwardThree compounds, A, B, and C, have the same molecular formula, C5H6. In the presence of a platinum catalyst, all three compounds absorb 3 molar equivalents of hydrogen and yield pentane. Compounds B and C give a precipitate when treated with ammoniacal silver nitrate; compound A give no reaction. Compounds A and B show an absorption maximum near 250 nm. Compound C shows no absorption maximum beyond 200 nm. Propose a structure for A, B, and C.arrow_forward
- Treatment of compound E (molecular formula C4H8O2) with excess CH3CH2MgBr yields compound F (molecular formula C6H14O) after protonation with H2O. E shows a strong absorption in its IR spectrum at 1743 cm-1. F shows a strong IR absorption at 3600–3200 cm-1. The 1H NMR spectral data of E and F are given. What are the structures of E and F?Compound E signals at 1.2 (triplet, 3 H), 2.0 (singlet, 3 H), and 4.1 (quartet, 2 H) ppmCompound F signals at 0.9 (triplet, 6 H), 1.1 (singlet, 3 H), 1.5 (quartet, 4 H), and 1.55 (singlet, 1 H) ppmarrow_forwardIdentify the structure of compound C (molecular formula C11H15NO2), which has an IR absorption at 1699 cm−1 and the 1H NMR spectrum shown below.arrow_forwardCompound A undergoes an acid-catalyzed hydrolysis. One of the products (B) that is isolated gives the following 1H NMR spectrum. Identify the compounds A and Carrow_forward
- Compounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forwardCompound A has molecular formula C8H8O. Its 1H-NMR spectrum shows a singlet of integral 3 at 2.58 ppm and a multiplet of integral 5 at 7.84 ppm. Its 13C-NMR shows six signals. The IR spectrum of A shows a noteworthy stretch at 1698 cm-1. - Identify Compound A, explaining your reasoning Compound A is treated with a mixture of nitric and sulfuric acids to generate Compound B. The 1H-NMR spectrum of B shows three singlets, one at 2.52 pm of integral 3, one at 6.67 ppm of integral 1, and one at 7.94 ppm of integral 2. The 13C-NMR spectrum of B shows six signals. - Identify compound B, explaining your reasoning Compound B is treated with methylmagnesium bromide and then aqueous acid to generate compound C. The 1H-NMR spectrum of C shows four singlets, one at 1.42 ppm of integral 6, one at 2.3 ppm of integral 1, one at 8.52 ppm of integral 2, and one at 8.97 ppm of integral 1. The 13C-NMR spectrum of C shows six signals. The IR spectrum of C shows a noteworthy broad stretch at 3326 cm-1. -…arrow_forwardTreatment of benzoic acid (C6H5CO2H) with NaOH followed by 1-iodo-3methylbutane forms H. H has a molecular ion at 192 and IR absorptions at 3064, 3035, 2960−2872, and 1721 cm−1. Propose a structure for H.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY