
(a)
Interpretation:
The mechanism of 4-Hydroxybenzaldehyde to compound A has to be shown.
(b)
Interpretation:
The reagent and experiment condition is to be given for the conversion of A to B.
Concept introduction:
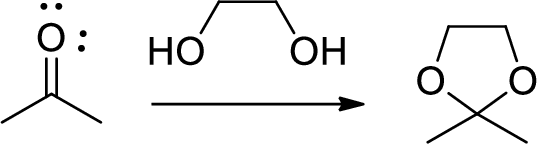
(c)
Interpretation:
The mechanism for the conversion of B to C is to be explained.
Concept introduction:
Wittig Reaction: It is an organic reaction where an aldehyde or a ketone gets converted to
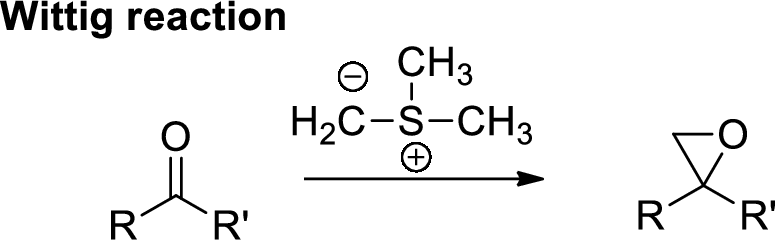
(d)
Interpretation:
The reagent and experiment condition is to be given for the conversion of C to D.
Concept introduction:
(e)
Interpretation:
The reagent and experiment condition is to be given for the conversion of D to albuterol.
Concept introduction:
Diol is to protect the ketone and aldehyde (carbonyl group). In this reaction acetone is protected as acetal by using ethylene glycol.
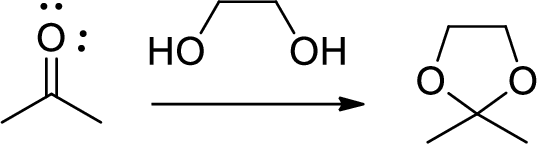
(f)
Interpretation:
The possible stereoisomer’s has to be shown if the product is chiral.
Concept introduction:
Chiral:
A molecule is non superimposable on its mirror image is called chiral molecule. Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Trending nowThis is a popular solution!

Chapter 21 Solutions
Organic Chemistry
- Show how the following ketones might be synthesized from the indicated acids, usingany necessary reagents.(b) methyl cyclohexyl ketone from cyclohexanecarboxylic acidarrow_forwardb) Propose a synthesis for the following compound from benzene. OCH3arrow_forward3) Draw equations of the following reactions and and explain to which direction is the respective quillibrium shifted. a) cyclohexylamine + water b) aniline + sulphuric acid c) triethylamine + acetic acidarrow_forward
- (b) Answer the following questions based on the compounds below. Jawab soalan berikut berdasarkan kepada sebatian di bawah. CI CI A в (i) Which compound has the higher boiling point? Explain. Sebatian manakah mempunyai takat didih yang lebih tinggi? Terangkan. (ii) Draw the SN2 mechanism for the reaction of compound A with sodium hydroxide, NaOH. Lukis mekanisma Sn2 bagi tindak balas antara sebatian A dengan natrium hidroksida, NaOH.arrow_forwardShow how to accomplish the following alkylation. (a) Step 1 (i) Specify the reagent used in this step. (ii) Draw the structure of the intermediate formed in this step, compound A. (B) draw the structure of the reagent needed for this step. (C) specify the reagent used in step b C Show how to accomplish the following alkylation. [References] Step 1 Step 2 compound A Step 3 CO₂Et (a) Step 1 (i) Specify the reagent used in this step. (ii) Draw the structure of the intermediate formed in this step, compound A. 1arrow_forwardKindly answer this questionarrow_forward
- 1) Carry out TWO of the following three synthesis. (You can do the third one for . Provide all the reagents and show all structures of the products formed in each step, not just a list of reagents. a) Benzene b) Benzene c) Toluene p-aminobenzoic acid OHC COOHarrow_forwardGive detailed Solution with explanation neededarrow_forwardShow how to synthesize the compound from benzene.arrow_forward
- Write a retrosynthesis and a foeard synthesis for the desired product. (c) OH ???arrow_forward(a) Explain why an alkylamine is more basic than ammonia?(b) How would you convert(i) Aniline to nitrobenzene (ii) Aniline to iodobenzenearrow_forwardGive me a clear handwritten answer with explanationarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
