
Concept explainers
(a)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given two steps to derive the corresponding structure of a
Answer to Problem 21.6P
The structure corresponding to the given name is,
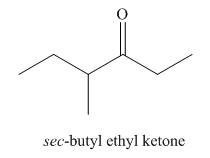
Explanation of Solution
The given name is sec-butyl ethyl ketone.
One should follow the given two steps to derive the corresponding structure of a ketone from its common name. The first step is identification of alkyl group that common name suggests. The second step is connecting these groups with
The given name suggests that sec-butyl and ethyl groups are bonded to
Thus, the structure corresponding to the given name is,

Figure 1
The structure corresponding to the given name is drawn in Figure 1.
(b)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given two steps to derive the corresponding structure of a ketone from its common name. The first step is identification of alkyl group that common name suggests. The second step is connecting these groups with
Answer to Problem 21.6P
The structure corresponding to the given name is,
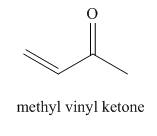
Explanation of Solution
The given name is methyl vinyl ketone.
One should follow the given two steps to derive the corresponding structure of a ketone from its common name. The first step is identification of alkyl group that common name suggests. The second step is connecting these groups with
The given name suggests that methyl and vinyl groups are bonded to
Thus, the structure corresponding to the given name is,

Figure 2
The structure corresponding to the given name is drawn in Figure 2.
(c)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given two steps to derive the corresponding structure of a ketone from its common name. The first step is identification of alkyl group that common name suggests. The second step is connecting these groups with
Answer to Problem 21.6P
The structure corresponding to the given name is,
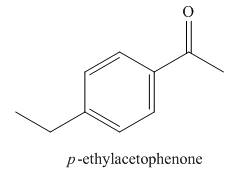
Explanation of Solution
The given name is p-ethylacetophenone.
One should follow the given two steps to derive the corresponding structure of a ketone from its common name. The first step is identification of alkyl group that common name suggests. The second step is connecting these groups with
The given name suggests the presence of acetophenone, whose structure is,
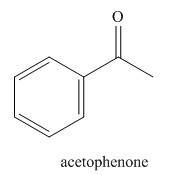
Figure 3
Since the ethyl group is present at para position, the structure corresponding to the given name is,

Figure 4
The structure corresponding to the given name is drawn in Figure 4.
(d)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given two steps to derive the corresponding structure of a cyclic ketone from its common name. The first step is identification of substituents bonded to it. The second step is connecting these groups at appropriate positions, by placing the carbon of
Answer to Problem 21.6P
The structure corresponding to the given name is,
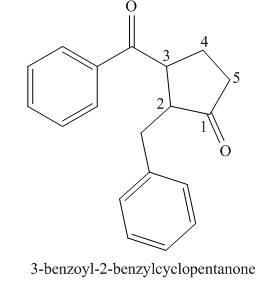
Explanation of Solution
The given name is
One should follow the given two steps to derive the corresponding structure of a cyclic ketone from its common name. The first step is identification of substituents bonded to it. The second step is connecting these groups at appropriate positions, by placing the carbon of
The given name suggests the presence of cyclic ring containing five carbon atoms, on which
Thus, the structure corresponding to the given name is,

Figure 5
The structure corresponding to the given name is drawn in Figure 5.
(e)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given two steps to derive the corresponding structure of a cyclic enone from its common name. The first step is identification of substituents bonded to it and position of double bond. The second step is connecting the groups at appropriate positions, by placing the carbon of
Answer to Problem 21.6P
The structure corresponding to the given name is,
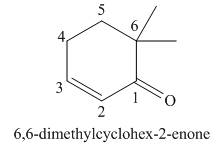
Explanation of Solution
The given name is
One should follow the given two steps to derive the corresponding structure of a cyclic enone from its common name. The first step is identification of substituents bonded to it and position of double bond. The second step is connecting the groups at appropriate positions, by placing the carbon of
The given name suggests the presence of cyclic ring containing six carbon atoms, on which
Thus, the structure corresponding to the given name is,

Figure 6
The structure corresponding to the given name is drawn in Figure 6.
(f)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given two steps to derive the corresponding structure of an enal (compound containing both
Answer to Problem 21.6P
The structure corresponding to the given name is,
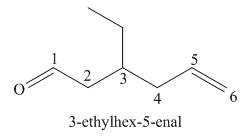
Explanation of Solution
The given name is
One should follow the given two steps to derive the corresponding structure of an enal (compound containing both aldehyde and a
The given name suggests the presence of longest chain containing six carbon atoms, on which
Thus, the structure corresponding to the given name is,

Figure 7
The structure corresponding to the given name is drawn in Figure 7.
Want to see more full solutions like this?
Chapter 21 Solutions
ORG.CHEMISTRY W/ACCESS+MODEL KIT PKG
- When (R)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH,nucleophilic substitution yields an optically inactive solution. When theisomeric halide (R)-2-bromo-2,5-dimethylnonane is dissolved in CH3OHunder the same conditions, nucleophilic substitution forms an opticallyactive solution. Draw the products formed in each reaction, and explainwhy the difference in optical activity is observed.arrow_forward2-Pentylcinnamaldehyde, commonly called flosal, is a perfume ingredient with a jasminelike odor. Flosal is an α,β-unsaturated aldehyde made by a crossed aldol reactionbetween benzaldehyde (C6H5CHO) and heptanal (CH3CH2CH2CH2CH2CH2CHO), followed by dehydration. Draw a stepwise mechanism for the following reaction that prepares flosal.arrow_forwardHydrogenation of alkene A with D2 in the presence of Pd-C affords asingle product B. Keeping this result in mind, what compound is formedwhen A is treated with each reagent: (a) mCPBA; (b) Br2, H2O followedby base? Explain these results.arrow_forward
- Could u show the products and the mechanism of the following reaction ( A=Hemiacetal, B= acetal) why is the same rection not forming a cyclic hemiacetal with 7-hydroxyheptanal ? How is the second reaction catalyzed? Want solution ASAp with detailed mechanismarrow_forwardcis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a different substitution product when they react with HO-. a. Why do they form the same elimination product? b. Explain, by showing the mechanisms, why different substitution products are obtained. c. How many stereoisomers does each of the elimination and substitution reactions form?arrow_forwardWhat reagents ( A–D) are needed to carry out each reaction in the following sequence?arrow_forward
- Why do you suppose ketone halogenations in acidic media are referred to as being acid-catalyzed, whereas halogenations in basic media are base-promoted? In other words, why is a full equivalent of base required for halogenation?arrow_forwardHydrogenation of alkene A with D2 in the presence of Pd-C affords a single product B. Keeping this result in mind, what compound is formed when A is treated with each reagent: (a) mCPBA; (b) Br2, H2O followed by base? Explain these results.arrow_forwardHow to synthesize the alkyl azide?arrow_forward
- a) Which reaction yields hexan-1-ol?arrow_forwardDraw and upload a reaction scheme to show how you would make butan-2-one starting from but-1-ene clearly showing products and needed reagents.Hint: multiple reactions are required!arrow_forwardβ-Vetivone is isolated from vetiver, a perennial grass that yields a variety of compounds used in traditional eastern medicine, pest control, and fragrance. In one synthesis, ketone A is converted to β-vetivone by a two-step process:Michael reaction, followed by intramolecular aldol reaction. (a) What Michael acceptor is needed for the conjugate addition? (See Problem 23.61 for another method to form the bicyclic ring system of β-vetivone.) (b) Draw a stepwise mechanism for the aldol reaction, which forms the six-membered ring.arrow_forward
