
(a)
Interpretation:
Electronic configuration of the following metals has to be written –
- (a) Ni (b) Cd (c) Zr (d) Os
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
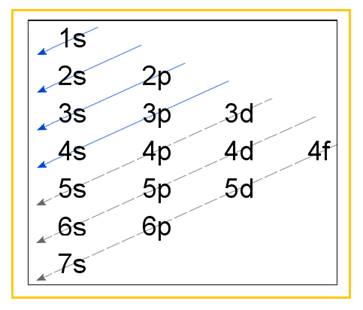
Figure 1
The terms
(b)
Interpretation:
Electronic configuration of the following metals has to be written –
- (a) Ni (b) Cd (c) Zr (d) Os
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
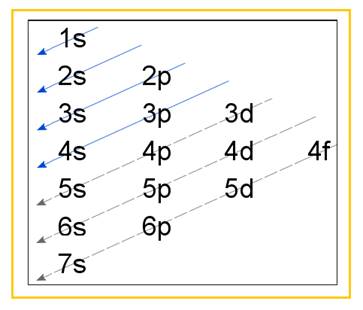
Figure 1
The terms
(c)
Interpretation:
Electronic configuration of the following metals has to be written –
- (a) Ni (b) Cd (c) Zr (d) Os
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
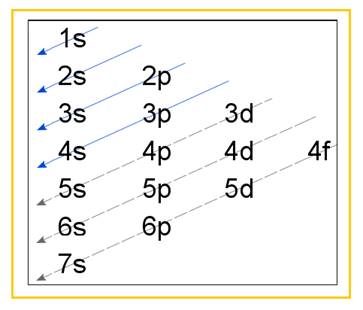
Figure 1
The terms
(d)
Interpretation:
Electronic configuration of the following metals has to be written –
- (a) Ni (b) Cd (c) Zr (d) Os
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
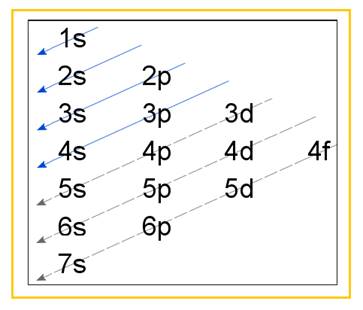
Figure 1
The terms
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Bundle: Chemistry, 9th + OWLv2 with MindTap Reader, 4 terms (24 months) Access Code
- The standard reduction potential for the reaction [Co( H 2 O)6]3+(aq)+e[CO( H 2 O)6]2+(aq) is about 1.8 V. The reduction potential for the reaction [Co( NH 3 )6]3+(aq)+e[Co( NH 3 )6]2+(aq) is +0.1 V. Calculate the cell potentials to show whether the complex ions,. [Co( H 2 O)6]2+ and or [Co( NH 3 )6]2+, can be oxidized to the corresponding Cobalt (III) complex by oxygen.arrow_forwardWrite an abbreviated orbital diagram and determine the number of unpaired electrons in each species in Question 31.arrow_forwardGive the oxidation state of the metal, number of d electrons, and the number of unpaired electrons predicted for [CO(NH3)6]Cl3.arrow_forward
- Chelating ligands often form more stable complex ions than the corresponding monodentate ligands with the same donor atoms. For example, Ni2+(aq)+6NH3(aq)Ni(NH3)62+(aq)K=3.2108Ni2+(aq)+3en(aq)Ni(en)32+(aq)K=1.61018Ni2+(aq)+penten(aq)Ni(penten)2+(aq)K=2.01019 where en is ethylenediamine and penten is This increased stability is called the chelate effect. Based on bond energies, would you expect the enthalpy changes for the above reactions to be very different? What is the order (from least favorable to most favorable) of the entropy changes for the above reactions? How do the values of the formation constants correlate with S? How can this be used to explain the chelate effect?arrow_forwardWhy do ligands cause the d orbitals of a transition metal ion to cease being degenerate? A. New molecular orbitals are formed via atomic orbital overlapt B. There is repulsion between the ligands and the d orbitals. C. They destabilize the ion nucleus D. Donation of electrons to the metal ion results in redox chemistryarrow_forwardWhich reagent is capable of oxidizing Cu to Cu2+ but does not oxidize Au to Au3+ ? a. More than one these b. Sn4+ c. Tl(s) d. I2(s) e. None of these f. Bi(s) g. IO3– h. MnO4–arrow_forward
- Which first-row transition metal(s) has the following highest possible oxidation state?a. +3 b. +7 c. +4arrow_forwarda. Fill in the energy level diagram for vanadium. b. Explain why some of the most common charge options (oxidation states) for vanadium are +2, +3, +4, +5. Increasing energy 75 6s 5s 45 3s 2s 1s 7p бр 5p 4p 3p 2p 6d 5d 4d 3d 5f 4f BYJU'S The Leaming Apparrow_forwardThe element which has the electronic configuration [Kr), 55, 4d is IS a. Paramagnetic in its atomic state b. Paramagnetic at oxidation state +2 c. Diamagnetic at its atomic state d. Diamagnetic colored substancearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





