
Concept explainers
a) p-Bromo phenyl acetamide.
Interpretation:
To draw the structural formula from the given name. We retrace steps as in the IUPAC system.
Answer to Problem 46AP
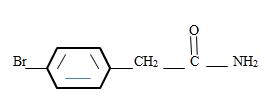
Explanation of Solution
To derive the structure corresponding to the given IUPAC name, we need to take recourse to the IUPAC system, but somewhat retracing the steps. Thus:
1) The given name p-Bromo phenyl acetamide than the suffix –amide.
At the very outset, Thus, the compound is invariably an amide.
2) The general formula for an amide is
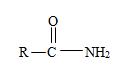
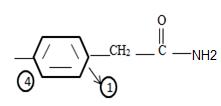
3) Identify now the alkyl group R attached to the carbonyl carbon. This, for the given name p-Bromo phenyl aceta is
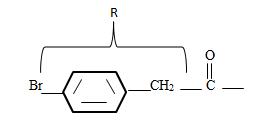
The phenyl ring with a Bromine for at the pare position curt the intervening –CH2. That is 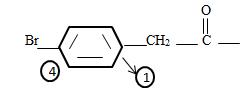
4) Recall the parent acetic acid is a 2-carbon acid system
It the given compound the phenyl ring is at C2 ie
It is a substituted acetic acid. Hence the aceta naming
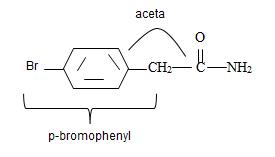
Combining these aspects, it follows that the parent carboxylic acid is acetic acid, substituted by the p-bromo phenyl at one H is 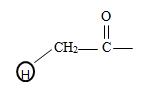 by
by
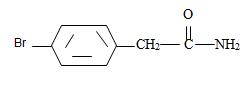
Thus the structure of the given compound where name is p-bromo phenyl acetamide is
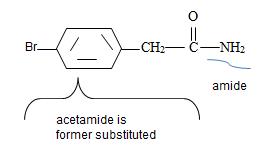
Is further substituted
Thus the structure corresponding to the given name p-bromo phenyl acetamide is
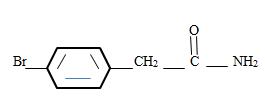
b) m-Benzoylbenzamide
Interpretation:
To draw the structural formula from the given name. We retrace steps as in the IUPAC system.
Answer to Problem 46AP
Explanation of Solution
The structure corresponding to the given name can be obtained by taking recourse to the IUPAC system, in a reverse way: Thus
1) First, the name ends with –amide as suffix. Thus, the compound is an amide and its general formula may be written
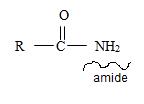
2) Now, identify the R ie alkyl group attached to the carbonyl carbon.
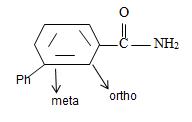
[Also, it must be noted that the amide is not substituted at nitrogen as the name has no N-alkyl prefix] the R is m-benzoyl benzoyl implying 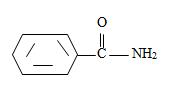 for Benzamide with a further benzoyl, at the meta carbon, C-3 of the phenyl ring.
for Benzamide with a further benzoyl, at the meta carbon, C-3 of the phenyl ring.
Thus,
or
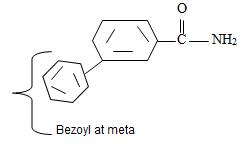
Thus the structure of the compounds as
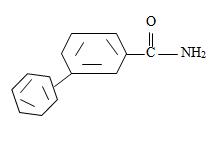
Thus the compound with the given IUPAC name m-benzoyl benzamide has the structure
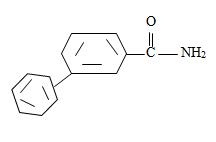
c) 2,2-Dimethyl hexanamide
Interpretation:
To draw the structural formula from the given name. We retrace steps as in the IUPAC system.
Answer to Problem 46AP
Explanation of Solution
The structure corresponding to the given IUPAC mane 2,2-Dimethylhexanamide can be obtained by retracing the steps of the IUPAC system. Thus,
a) First, observe that the given, IUPAC name has the suffix–amide.
Thus the compound is an amide.
b) The name has no alkyl substituent at the nitrogen of the amino –NH2 group. So the compound should have a structure consistent with the general formula
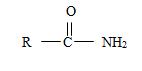
c) Now to identify the R-alkyl portion, we see it is 2,2-Dimethyl hexane chain, ie
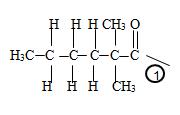
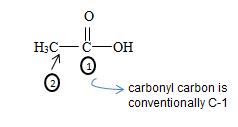
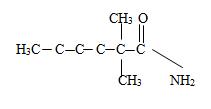
By convention, the carbonyl carbon is C-1, C-2 has 2 methyl groups. Thus the parent (longest) hydrocarbon chain –R is 2,2-dimethylhexane.
Thus the structure should be
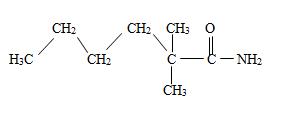
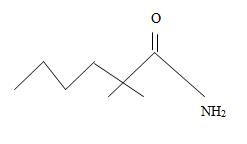
Thus the structure corresponding to the compound whose IUPAC name is 2,2-dimethyl hexanamide is
d) Cyclohexyl cyclohexane carboxylate
Interpretation:
To draw the structural formula from the given name. We retrace steps as in the IUPAC system.
Answer to Problem 46AP
Explanation of Solution
The structure of the compound with the given IUPAC name
Cyclohexyl cyclohexane carboxylate can be obtained by retracing the rules of the IUPAC system: Thus.
1) First the, given name has its suffix: carboxylate
Thus it is an ester of a carboxylic acid.
2) Second, recall that the alkyl R1 of the ester oxygen comes first in the IUPAC name. In the given name this R1 is cyclohexyl. Thus the OR1 of the ester (general)
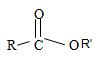
Is cyclohexyl structure is
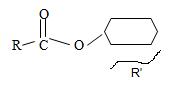
3) For the R (alkyl) component attached to the carbonyl carbon, it is given as cyclohexane. This implies the structure
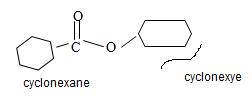
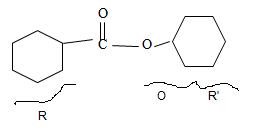
By convention, the cyclohexane carboxylic acid ester is named by replacing the carboxylic acid ending by carboxylate. Assembling all these rules, the structure of the compound corresponding to the name cyclohexyl cyclohexane carboxylate is
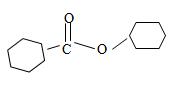
Thus the structure of the compound that best fits the given IUPAC name is
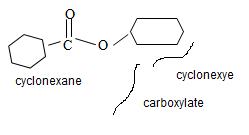
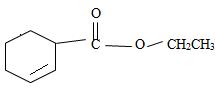
e) Ethyl-2-cyclobutene carboxylate
Interpretation:
To draw the structural formula from the given name. We retrace steps as in the IUPAC system.
Answer to Problem 46AP
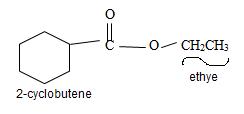
Explanation of Solution
The structure of the compound whose IUPAC name is Ethyl 2-cyclobutene carboxyl may be obtained by retracing the rules of the IUPAC system. Thus,
1) First, we see that the suffix of the name is carboxylate.
Thus the compound is an ester.
2) Second, the general formula of the ester may be written
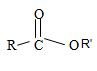
3) Third, by convention, the ester alkyl function R1 comes as prefix in the IUPAC name for the given compound it is ethyl –CH2CH3. Thus, the compound could be
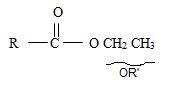
4) Next we identify that the alkyl component R of the carbonyl carbon from the name is 2-cyclobutene ie
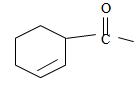
With the double bond at C-2; since C-1 of the cyclobutene ring must by convention carry the carboxylic carbon of the acyl group
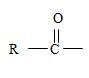
Finally the ester ending is carboxylate which agrees with the facts-is the compound is an ester of a cyclohexyl carboxylic acid
Thus the compound is an ester.
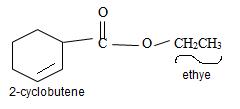
Thus the structure of the compound where IUPAC name is ethyl 2-cyclobutene carboxylate is
f) Succinic anhydride
Interpretation:
To draw the structural formula from the given name. We retrace steps as in the IUPAC system.
Answer to Problem 46AP
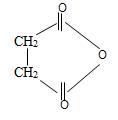
Explanation of Solution
The structured formula of the compound corresponding to the given name succinic an hydride is obtained by retracing the rules of the IUPAC system. Thus,
1) First, the given name has its suffix anhydride. The compound is thus, an anhydride of a carboxylic acid.
2) The general formula for an acid anhydride of a mono carboxylic acid as
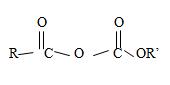
Where R and R1 are the two alkyl groups may be similar R=R in which care it is a synmetical an hydride, or R≠R1, for an unsynmetical an hydride. It is now needed to identify R and R1.
Now for this, we introspect the prefix of the name is succinic this implies the parent acid is a dicarboxylic acid is succinic acid. Its formula is
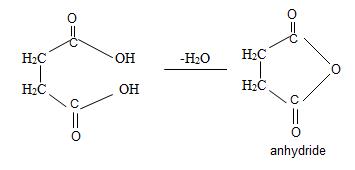
Since by reaction and convention, acid an hydride form by lon of a water molecule, thus,
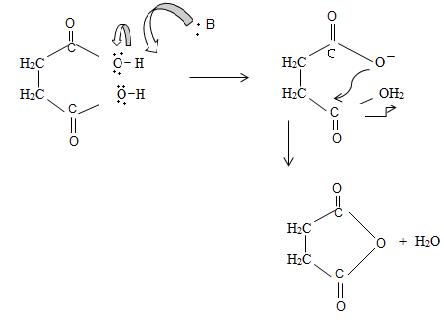
Thus consistent with the above facts the structural formula for succinic an hydride is
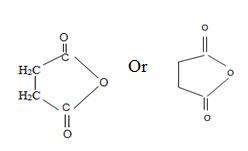
Thus the structural formula for succinic anhydride is

Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry - OWLv2 Access
- Several compounds have been found to inhibit -lactamase, and drugs based on these compounds can be taken in combination with penicillins and cephalosporins to restore their effectiveness when resistance is known to be a problem. The commonly prescribed formulation called Augmentin is a combination of the -lactamase inhibitor shown below with amoxicillin (shown above). It is used to treat childhood ear infections when resistance is suspected, and many kids know it as the white liquid that tastes like bananas. Which of the statements below are true statements? 1. The stereochemistry of the fusion between the four-and five-membered rings in the inhibitor and amoxicillin are different. 2. The inhibitor possesses enol ether and allylic alcohol functional groups while the antibiotic possesses a phenol and a secondary amide functional group. 3. Neither the inhibitor nor the antibiotic contains strained rings. 4. Both 1 and 2 are true.arrow_forwardFollowing is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker with high selectivity for airway smooth muscle receptors. Q.Suggest a structural relationship between carbuterol and ephedrinearrow_forward1. Biogenic Amines are inactivated primarily by a. Sulfonation b. Glucoronidation c. Aromatic hydroxylation d. Methylation 2. A drug can exert its pharmacological effect only if it is a. Protein bound b. Protein unbound c. Free drug d. Both B & C e. Both A & C 3. In order for the drug to be ready and available for absorption, it must be release first from its dosage form with the exception of: a. Capsule b. Tablet c. Solution d. Suspension 4. All of the following are true, except a. Solubility increase with decrease particle size b. Solubility increase with increase surface area c. Solubility increase with increase particle size d. Solubility decrease with decrease surface area 5. The rate in which the drug appears in the bloodstream is also known as a. Half-life b. Potency c. Bioavailability d. Area under the curvearrow_forward
- ripelennamine is a histamine H1 antagonist with low sedative action but frequent gastrointestinal irritation. It is used to treat asthma; hay fever; urticaria; and rhinitis. Based on biotransformations discussed in class, draw the structures of 3 possible metabolites of tripelennamine.arrow_forwardThe following three derivatives of succinimide are anticonvulsants that have found use in the treatment of epilepsy, particularly petit mal seizures. Q. Show how this same synthetic strategy can be used to prepare ethosuximide and methsuximide.arrow_forwardPropose a mechanism for the hydrolysis of N,N-dimethylacetamide b) under acidic conditions.arrow_forward
- Indicate whether the following statement is true or false . Benzenesulfonyl chloride or p-toluenesulfonylchloride give N-substituted sulfonamides with primary and secondary amines. The derivative consisting of primary amines is insoluble in dilute NaOH.RightFalsearrow_forwardPropose a mechanism for the reaction of glucosamine with acetylacetone to form product containing the pyrrole ring. Hint: The likely first step involves attack of the nucleophilic amino group of glucosamine on one of the carbonyl carbons of acetylacetone and leads to formation of an aminoalcohol. This can dehydrate to form a α,β unsaturated carbonyl compound. The alkene can then react with the aldehyde to form a ring structure.arrow_forwardPredict the chemical name of compound B a. p-chlorobenzylamine b. 4-bromoaniline c. benzylamine d. p-chloronitrile e. p-chlorobenzaldehyde 2. What is the reaction name for the chemical transformation of A to B a. reductive amination b. catalytic reduction c. carbonyl dehydration d. Hofmann elimination e. Aldehyde rearrangementarrow_forward
- d-Glucuronic acid is found widely in plants and animals. One of its functions is to detoxify poisonous HO-containing compoundsby reacting with them in the liver to form glucuronides. Glucuronides are water soluble and therefore readily excreted. Afteringestion of a poison such as turpentine or phenol, the glucuronides of these compounds are found in the urine. Draw thestructure of the a- and b-glucuronides formed by the reaction of b-d-glucuronic acid and phenol.arrow_forwardExplain the mechanism for the acid catalyzed hydrolysis of an amide?arrow_forwardExplain Imine and Enamine Hydrolysis ?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

