
Concept explainers
(a)
Interpretation:
The curved arrow mechanism, for base catalyzed exchange of hydrogen
Concept introduction:
A carbonyl group has
Answer to Problem 22.74AP
The curved arrow mechanism for base catalyzed exchange of hydrogen
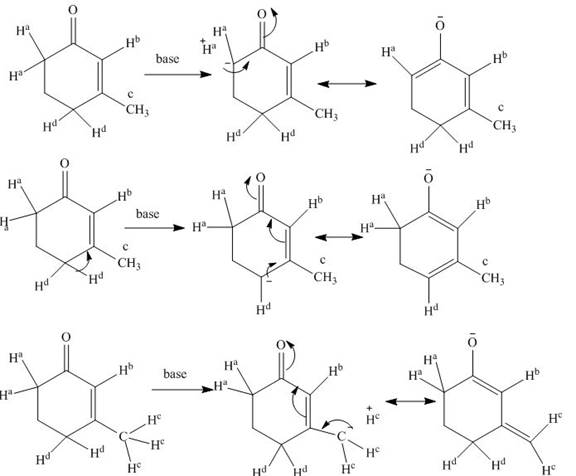
Explanation of Solution
The structure of compound is given below.
Figure 1
When a base is present in the medium, it easily removes the
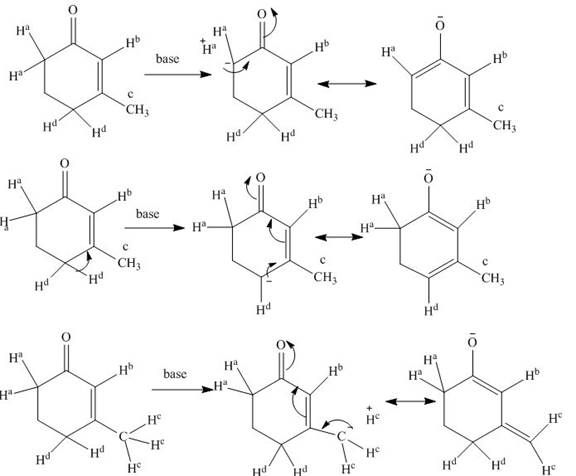
Figure 2
The base catalyzed exchange of hydrogen
(b)
Interpretation:
The less acidic character of
Concept introduction:
The
Answer to Problem 22.74AP
The proton,
Explanation of Solution
The
Figure 3
The
(c)
Interpretation:
The mechanism for exchange of
Concept introduction:
In presence of the acid or base, the enone compounds remain in equilibrium with their isomers. Due to this, acidity of
Answer to Problem 22.74AP
The isomer
Explanation of Solution
The equilibrium of isomers,
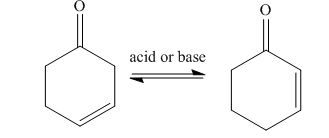
Figure 4
The
The reaction mechanism is shown in figure below.
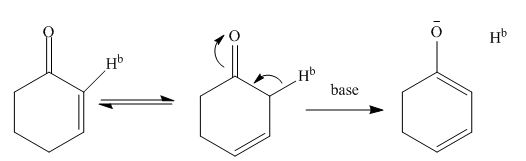
Figure 5
The
(d)
Interpretation:
The hydrogen that is replaced by deuterium in sex hormone, testosterone is to be identified.
Concept introduction:
The
Answer to Problem 22.74AP
The hydrogen at position “a” in vicinity to carbonyl carbon can be exchanged for deuterium in
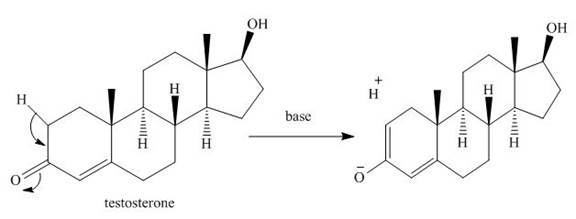
Explanation of Solution
The structure of sex hormone testosterone is given below.
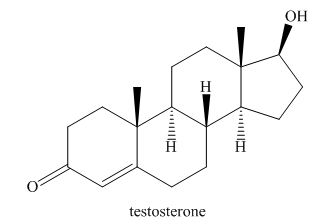
Figure 6
The hydrogen present at position “a” can be exchanged with deuterium, in sex hormone testosterone. This hydrogen, being at
The mechanism for the same can be shown as given below.

Figure 7
The
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry Study Guide and Solutions
- a) Rank the following compounds from most basic to least basic. HC C A e H3C-CH₂ B b) Clearly explain the rationale behind your answer. H₂C=CH C HC=CH Ꭰarrow_forwardProvide structures A, B and Carrow_forwardArrange these compounds in order of increasing boiling point (values in °C are -42, -24, 78, and 118). (a) CH,CH,OH (b) CH,OCH, (c) CH,CH,CH, (d) CH,COOHarrow_forward
- Rank the following acids in decreasing acidity. Explain your answer CH2ClCOOH, CHCl2COOH, CH3COOHarrow_forward16arrow_forward1.(a) Which of the following groups has the LOWEST IUPAC priority?(A) CH3 (B) NH2 (C) OH (D) COOH (E) Br (b)Which of the following corresponds to the strongest acid?(A) (CF3)3C-COOH (B) (CF3)3 C-OH(C) CH3COOH (D) CH3OH(E) HOCH2CH3arrow_forward
- b) Rank the following molecules according to their acidity. Justify your answer. O OH NO₂ O OH CH 3 O OH CIarrow_forwardCompound A exhibits a peak in its 1H NMR spectrum at 7.6 ppm, indicating that it is aromatic. (a) How are the carbon atoms of the triple bonds hybridized? (b) In what type of orbitals are the π electrons of the triple bonds contained? (c) How many π electrons are delocalized around the ring in A?arrow_forwardwhich of the following compound shows the strongest acidity A-ch3oh B-ch3ch2oh C-h2o D-c6h5oh 1-A 2-B 3-C 4-D 5-Earrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

