
Concept explainers
(a)
Interpretation: The product that is formed by the reaction of
Concept introduction: The bimolecular elimination reaction that favours the removal of a HX substituent from an
Answer to Problem 23.13P
The product that is formed by the reaction of
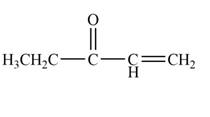
Explanation of Solution
The product that is formed by the reaction of
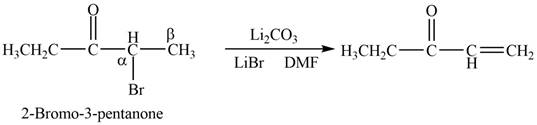
Figure 1
In this reaction, the given
The product that is formed by the reaction of
(b)
Interpretation: The product that is formed by the reaction of
Concept introduction: The substitution reaction involves the replacement of one functional group by other functional group. In nucleophilic substitution an electron rich species attack the species that is deficient in electrons. The electrophile and the leaving group together form a substrate. The nucleophile attacks over the substrate and there occurs the removal of leaving group from the substrate.
The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 23.13P
The product that is formed by the reaction of
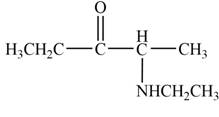
Explanation of Solution
The product that is formed by the reaction of
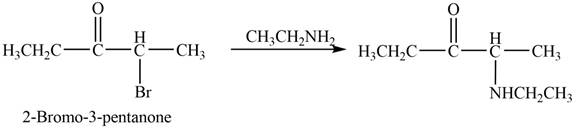
Figure 2
In this reaction, the given ketone,
The product that is formed by the reaction of
(c)
Interpretation: The product that is formed by the reaction of
Concept introduction: The substitution reaction involves the replacement of one functional group by other functional group. In nucleophilic substitution an electron rich species attack the species that is deficient in electrons. The electrophile and the leaving group together form a substrate. The nucleophile attacks over the substrate and there occurs the removal of leaving group from the substrate.
The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 23.13P
The product that is formed by the reaction of
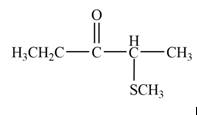
Explanation of Solution
The product that is formed by the reaction of
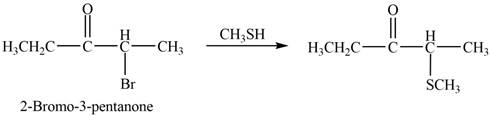
Figure 3
In this reaction, the given ketone,
The product that is formed by the reaction of
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY-STUDY GUIDE PACKAGE
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardCH3-CH2-OH reacts with H+/140C = ? + H2Oarrow_forwarda) Write the name b) draw the structure product formed from the reaction of 3-methylpent-2-ene with [1] 9-BBN; [2] H2O2, OH-arrow_forward
- Draw the product formed when C6H5N2+Cl− reacts with each compound.arrow_forwardDraw the product formed when C6H5N2 +Cl− reacts with each compound.arrow_forward3. a. What is the chemical structure of benzoic acid, circle functional groups different than alkane,alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________arrow_forward
- Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forwarda. What is the chemical structure of 2,6-dichloroindophenol, circle functional groups differentthan alkane, alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? ___________arrow_forwardWhat happens when (i) Chlorobenzene is treated with Cl2/FeCl3 (ii) ethyl chloride is treated with AgN02, (iii) 2-bromopentane is treated with alcoholic KOH? Write the chemical equations in support of your answer.arrow_forward
- draw structure for 4-bromo-2-pentene where the double bond has a Z geometry and the chiral carbon an S configuration ?arrow_forwardCan acetone react with sodium bicarbonate (NaHCO3)? Why or why not?arrow_forward4. Write the formula and name for the product when cyclopentene reacts with (a) Cl2(b) HBr(c) H2, Pt (d) H2O, H+arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




