
Concept explainers
(a)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted
- (1) Name the parent alkane (long alkyl chain)
- (2) Number the carbon
- (3) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
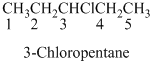
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
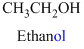
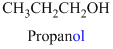
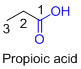
The given compound is an acid (
The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
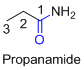
If the molecule is ester,
Esters end with “ate”
Example
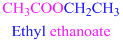
The given compound is an
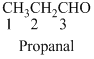
The given compound is an aldehyde (
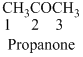
The given compound is an aldehyde (
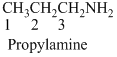
(a)
Answer to Problem 23.20QP
Answer
Structure of the given organic compounds is shown below (a).
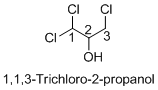
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is 1, 1, 3-trichloro-2-propanol.
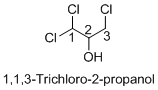
Parent chain is identified and numbering is given for the compound. According to the name, first carbon is bearing two chlorine groups and third carbon is bearing one chlorine group and second carbon is bearing hydroxyl group. The given name is propane which clearly indicates that the compound having three carbon atoms in the molecule. According to the name, the structure of the molecule is given above.
(b)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (4) Name the parent alkane (long alkyl chain)
- (5) Number the carbon
- (6) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
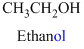
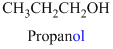
The given compound is an acid (

The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
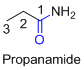
If the molecule is ester,
Esters end with “ate”
Example
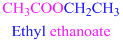
The given compound is an aldehyde (
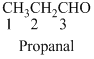
The given compound is an aldehyde (
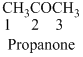
The given compound is an aldehyde (
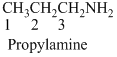
(b)
Answer to Problem 23.20QP
Answer
Structure of the given organic compounds is shown below (b).
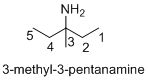
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is 3-methyl-3-pentanamine.
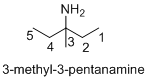
Parent chain is identified and numbering is given for the compound. According to the name, third carbon is bearing methyl group and amine group and the parent name of the molecule is pentane which clearly indicates that the compound having five carbon atoms in the parent. According to the name, the structure of the molecule is given above.
(c)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (7) Name the parent alkane (long alkyl chain)
- (8) Number the carbon
- (9) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
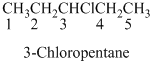
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
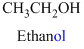
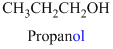
The given compound is an acid (

The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
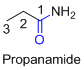
If the molecule is ester,
Esters end with “ate”
Example
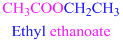
The given compound is an aldehyde (
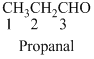
The given compound is an aldehyde (
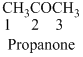
The given compound is an aldehyde (
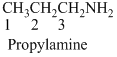
(c)
Answer to Problem 23.20QP
Structure of the given organic compounds is shown below (c).
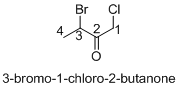
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is 3-bromo-1-chloro-2-butanone.
Parent chain is identified and numbering is given for the compound. According to the name, first carbon is bearing chlorine atom and third carbon is bearing bromine atom, second carbon is carbonyl carbon and the parent name of the molecule is butane which clearly indicates that the compound having four carbon atoms in the parent. According to the name, the structure of the molecule is given above.
(d)
Interpretation:
The structure of the organic compounds should be given.
Concept introduction:
Organic compounds are named systematically by using IUPAC rules.
Name of the organic compounds are given according to the number of carbon present in the molecule for example
A molecule having one carbon atom, the molecule name will start with meth etc.…

If any halogens are present in the molecule, the name of the halogens as follows.

Naming the substituted alkane:
- (10) Name the parent alkane (long alkyl chain)
- (11) Number the carbon
- (12) Name and number the substituent
If the molecules have the multiple substituents, the compound named as di, tri, tetra, penta, ect.
If the molecules having functional group, the name of the compound is given below. Numbering should be starts from the functional group of the given molecule.
The given compound is an alcohol
Example is given below
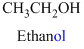
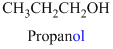
The given compound is an acid (

The amides are derivatives of acids and it is named as the ending of alkane with amide.
For example
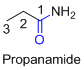
If the molecule is ester,
Esters end with “ate”
Example
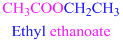
The given compound is an aldehyde (
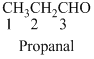
The given compound is an aldehyde (
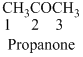
The given compound is an aldehyde (
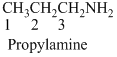
(d)
Answer to Problem 23.20QP
Structure of the given organic compounds is shown below (d).
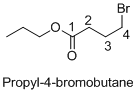
Explanation of Solution
To find: The structure of the given organic molecule.
Name of the given organic compounds is propyl-4-bromobutanoate.
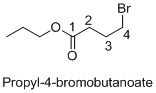
Parent chain is identified and numbering is given for the compound. According to the name, fourth carbon is bearing bromine atom, the given molecule is ester so propyl group is attached after oxygen atom in the molecule and the parent name of the molecule is butane which clearly indicates that the compound having four carbon atoms in the parent. According to the name, the structure of the molecule is given above.
Want to see more full solutions like this?
Chapter 23 Solutions
Loose Leaf For Chemistry: Atoms First With Connect 2y Access Card
- draw the structure of C8H10NO7P2- and give the general name of the functional grouparrow_forwardWrite the chemical equation for the reaction of propanoic acid with 1-butanol (an alcohol). The formula of 1-butanol is CH-CH-CH2-CH2-OH. Which is formula of the this rule of reaction?arrow_forwardWrite structures and describe thephysical properties of carboxylic acidsarrow_forward
- Write the structure of the following compounds: a. cyclohexyl propanoate, b. 4-methylheptanonitrile, c. butanoic benzoic anhydride, d. N,N-dibenzylmethamide, e. 3-methylhexanoyl chloride.arrow_forwardWrite the structures of the following compounds. a) N-methyl-o-toluidine b) 3-Benzyl-5-ethylheptanoic acidarrow_forwardWrite a condensed structural formula for a dihydroxy compound with the formula C3H8O2.arrow_forward
- write the structure formulas of alkanes with molecular formula C6H14, which with chlorine give: a) three monochlorinated isomers? b) five monochlorinated isomers c) only two monochlorinated isomersarrow_forwardWrite the structural formula of 2-ethyl-1-butenearrow_forwardAn isomer of C6H12O could contain a carboxylic acid. True or false?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



