
Concept explainers
(a)
Interpretation:
Molecular formula for the given set of compounds has to be written.
Concept Introduction:
Molecular formula of a compound represents the total number of atoms that is present in it. It does not give any other information about the functional group, etc present in the compound. Simply it gives how many number of atoms are present for each element that is present in the compound.
To Write: The molecular formula for the given ball-stick models.
(a)
Answer to Problem 23.23QP
The molecular formula for the first structure is
The molecular formula for the second structure is
The molecular formula for the third structure is
The molecular formula for the fourth structure is
Explanation of Solution
For First structure:
In the given ball-stick model, the black spheres are carbon atoms, light blue spheres are hydrogen atoms and dark blue spheres are nitrogen atoms. The total number of black spheres is 3, while that of light blue spheres is 9 and dark blue spheres is 1. Hence we arrive at the molecular formula of
For Second structure:
In the given ball-stick model, the black spheres are carbon atoms, light blue spheres are hydrogen atoms and red spheres are oxygen atoms. The total number of black spheres is 2, while that of light blue spheres is 4 and red spheres is 1. Hence we arrive at the molecular formula of
For Third structure:
In the given ball-stick model, the black spheres are carbon atoms, light blue spheres are hydrogen atoms and red spheres are oxygen atoms. The total number of black spheres is 3, while that of light blue spheres is 8 and red spheres is 1. Hence we arrive at the molecular formula of
For Fourth structure:
In the given ball-stick model, the black spheres are carbon atoms, light blue spheres are hydrogen atoms and red spheres are oxygen atoms. The total number of black spheres is 2, while that of light blue spheres is 4 and red spheres is 2. Hence we arrive at the molecular formula of
The molecular formula for the given ball-stick model compounds are written.
(b)
Interpretation:
Condensed structural formula for the given ball-stick model compounds has to be written.
Concept Introduction:
Condensed structural formula is representation of the organic compound. In this the lengthy carbon chain is shown only with the carbon atoms (along with the hydrogen) without any bonds. The additional branches are shown with explicit bonds.
To Write: The condensed structural formula for the given set of ball-stick model compounds.
(b)
Answer to Problem 23.23QP
The condensed structural formula of First structure is
The condensed structural formula for the Second structure is,
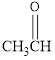
The condensed structural formula for the Third structure is,
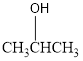
The condensed structural formula for the Fourth structure is,

Explanation of Solution
For First structure:
Condensed structural formula can be drawn considering the skeleton in the given ball-stick model. Nitrogen is the central atom which is bonded to three methyl groups. The single bonds between the nitrogen and methyl group are removed to obtain the condensed structural formula as shown below,

For Second structure:
Condensed structural formula can be drawn considering the carbon skeleton in the given ball-stick model. The carbon chain is drawn. One of the carbon atom is attached to an oxygen atom and a hydrogen atom. The single bonds are removed in the parent structure which results in the condensed structural formula as shown below,

For Third structure:
Condensed structural formula can be drawn considering the carbon skeleton in the given ball-stick model. The carbon chain is drawn. The carbon atom in the middle is attached to an hydroxy group. The single bonds are removed in the parent structure which results in the condensed structural formula as shown below,

For Fourth structure:
Condensed structural formula can be drawn considering the carbon skeleton in the given ball-stick model. The carbon chain is drawn. One of the carbon atom is attached to an hydroxy group and an oxygen atom. This means it is an carboxylic acid. The single bonds are removed in the parent structure which results in the condensed structural formula as shown below,

The condensed structural formula are written for the given ball-stick model representation of compounds.
(c)
Interpretation:
The functional group present in the given set of ball-stick models has to be identified.
Concept Introduction:
In an organic compound, the reactive portion is known as functional group. This undergoes reactions with other reagents and this does not depend upon how the rest of the compound is like. Few of the common functional groups are alcohol, ester, carboxylic acid,
To Identify: The functional group present in the given ball-stick models
(c)
Answer to Problem 23.23QP
The functional group present in First structure is identified as
The functional group present in Second structure is identified as aldehyde.
The functional group present in Third structure is identified as alcohol.
The functional group present in Fourth structure is identified as carboxylic acid.
Explanation of Solution
For First structure:
By looking at the formula of the given compound we can identify that a nitrogen is present in it. If nitrogen is present in a compound we can classify it as amine. In this case the nitrogen does not have any hydrogen atom bonded to it. Hence, it is a tertiary amine. The functional group is highlighted in red colour as shown.
For Second structure:
By looking at the formula of the given compound we can identify that a carbonyl group is present in it. The carbonyl group is attached to a hydrogen atom on one side and an alkyl group on other. Hence, this compound falls under the category of aldehyde.
For Third structure:
By looking at the formula of the given compound we can identify that a hydroxy group is present in it. The hydroxy group is attached to a carbon atom which bears only one hydrogen atoms. Hence, this compound falls under the category of secondary alcohol.
For Fourth structure:
By looking at the formula of the given compound we can identify that a carboxylic acid group is present in it. The carboxylic acid group is the functional group in this compound. Therefore, the compound falls under the category of carboxylic acid.
The functional groups present in the given set of ball-stick model compounds are identified.
Want to see more full solutions like this?
Chapter 23 Solutions
Bundle: General Chemistry, Loose-leaf Version, 11th + Lms Integrated For Owlv2 With Mindtap Reader, 4 Terms (24 Months) Printed Access Card
- Classifying the Carbons and Hydrogens in a Molecule Classify the designated carbon atoms in A as 1°, 2°, 3°, or 4°. Classify the designated hydrogen atoms in B as 1°, 2°, or 3°.arrow_forwardThe molecular formula of the molecule below is: Choices: A. C8H10B. C7H8C. C7H10D. C8H8arrow_forwardWhat are constitutional isomers? molecules with the same molecular formula but one triple bond instead of two double bonds molecules with the same structure but different substitutes molecules with the same structural formula but different spatial arrangements of atoms molecules with the same molecular formula but different structuresarrow_forward
- Tell how many hydrogens are bonded to each carbon in the following compounds, andgive the molecular formula of each substance:arrow_forwardDraw a structural formula for the compound with the molecular formula C8H12arrow_forwardMolecular formula. Molar mass (g.mol-1). ΔcH0 (kJ.mol-1) Petrol C8H18 * 114 - 5470 Diesel C20H42 * 282 - 8090 Natural gas. CH4 16 - 890 Hydrogen. H2 2 - 286 Methanol. CH3OH 32 - 726 *Petrol and diesel are complex mixtures of hydrocarbons. On average, their properties resemble those of C8H18 and C20H42, respectively. 1.1) Calculate the energy density (in kJ.g-1) for all the fuels in the table above. Use the molar masses supplied in the table. 1.2) Why would you say that Space Shuttles rather use hydrogen as its primary fuel source instead of one of the other fuels in the table? 1.3) Formula 1 racing cars use petrol as their fuel source. In the USA, “Indy cars” use methanol as fuel source and in South Africa farmers use diesel as fuel source for their tractors. If all these motors perform equally optimally, which vehicle is the most fuel efficient? Give a reason for your answer.arrow_forward
- CONSIDER THE COMPOUNDS BELOWarrow_forwardWhat is the chemical formula for the molecule represented by the skeletal structure below? = Select one: a. Cannot be determined from this skeletal structure b. C2 c. C2H2 d. CH4arrow_forwardDetermine the compound (name or structure) from the data. Explain features from each data Molecular formula: C3H8Oarrow_forward
- Which is true regarding the C-C bond lengths in benzene and cyclohexane? a benzene C-C length > cyclohexane C-C length b benzene C-C length = cyclohexane C-C length c benzene C-C length < cyclohexane C-C length d more info neededarrow_forwardWhat is the meaning of the term tertiary (3) when it is used to classify alcohols? Draw a structural formula for the one tertiary (3) alcohol with the molecular formula C4H10O.arrow_forwardDraw the structure of an alkane or cycloalkane that has more than three but fewer than ten carbon atoms, and only primary hydrogens. (There are several possible structures. It is enough to draw any one of them, but you may draw two or more if you want to.) Draw the unknown structure(s).arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





