
Concept explainers
(III) The dipole moment, considered as a vector, points from the negative to the positive charge. The water molecule, Fig. 23–32, has a dipole moment
where p is the magnitude or the net dipole moment,
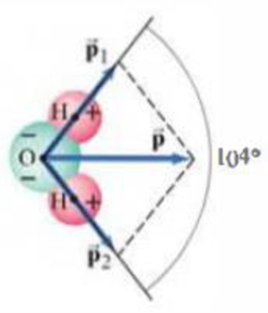
FIGURE 23–32 Problem 46.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
PHYSICS F/SCI.+ENGR.,CHAPTERS 1-37
Additional Science Textbook Solutions
Introduction to Electrodynamics
College Physics: A Strategic Approach (3rd Edition)
University Physics Volume 1
College Physics: A Strategic Approach (4th Edition)
An Introduction to Thermal Physics
- Calculate the angular velocity of an electron orbiting a proton in the hydrogen atom, given the radius of the orbit is 0.5301010 m. You may assume that the proton is stationary and the centripetal force is supplied by Coulomb attraction.arrow_forwardThree identical conducting spheres are fixed along a single line. The middle sphere is equidistant from the other two so that the center-to-center distance between the middle sphere and either of the other two is 0.125 m. Initially, only the middle sphere is charged, with qmiddle = +35.6 nC. The middle sphere is later connected by a conducting wire to the sphere on the left. The wire is removed and then used to connect the middle sphere to the sphere on the right. The wire is again removed. a. C What is the charge on each sphere? b. C Which sphere experiences the greatest electrostatic force? c. N What is the magnitude of that force?arrow_forwardA water molecule is made up of two hydrogen atoms and one oxygen atom, with a total of 10 electrons and 10 protons. The molecule is modeled as a dipole with an effective separation d = 3.9 1012 m between its positive and negative charges. What is the magnitude of the water molecules dipole moment?arrow_forward
- How many coulombs of positive charge are there in 4.00 kg of plutonium given its atomic mass is 244 and that each plutonium atom has 94 protons?arrow_forwardA water molecule consists of two hydrogen atoms bonded with one oxygen atom. The bond angle between the two hydrogen atoms is 104( (see below). Calculate the net dipole moment of a water molecule that is placed in a uniform, horizontal electric field of magnitude 2.3108N/C . (You are missing some information for solving this problem; you will need to determine what information you need, and look it up.)arrow_forwardWhat is the force on a dipole of dipole moment p placed as shown in the Fig. +9 a Fig. P -- +9arrow_forward
- a molecules of oxygen passess a charges of -2C and 2C separated from a distance of 0.2m. Calculate the dipole moment in this conditionarrow_forward(i) Derive the expression for electric field at a point on the equatorial line of an electric dipole. (ii) Depict the orientation of the dipole in (a) stable, (b) unstable equilibrium in a uniform electric field.arrow_forwardThe magnitude of the electric fie ld at a distance r from a point charge O is equal to O/4πεor2. How close to a water molecule (of polarizability volume 1.48 x 1-30 m3) must a proton approach before the dipole moment it induces is equal to the permanent dipole moment of the molecule (1.85 D)?arrow_forward
- Calculate the magnitude and direction of the dipole moment of the following arrangement of charges in the xy-plane: 4e at (0, 0), −2e at (162 pm, 0), and −2e at an angle of 30° from the x-axis and a distance of 143 pm from the origin.arrow_forwardSuppose the exact charge neutrality does not hold in a world and the electron has a charge 1% less in magnitude than the proton. Calculate the Coulomb force acting between two blocks of iron each of mass 1 kg separated by a distance of 1 m. Number of protons in an iron atom 26 = 26 and 58 kg of iron contains 6x 10 * atoms.arrow_forwardTwo opposite and equal charges 4 × 10-8 C when placed 2 × 10-² cm away, form a dipole. If this dipole is placed in -2 an external electric field 4×108 N/C, the value of maximum torque and the work done in rotating it through 180° will bearrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning


